New Method can Investigate Mechanical Forces in Live Tissue
To construct new tissue or maintain it in adulthood, cells have to be able to respond to mechanical and molecular cues from their environment and inform decisions like whether to become a nerve or skin or heart cell. Publishing in Nature Methods, scientists at UC Santa Barbara have created a method to observe the mechanical pressures that affect cells living in their natural and three-dimensional environment, discussed by the researchers in the following video.
"Knowing how cells respond to mechanical cues in the living embryo and how they physically sculpt tissues and organs in the 3D space will transform the way we think about developmental processes," said senior author Otger Campàs, a Mechanical Engineering Professor at UCSB. "Importantly, this knowledge will help us better understand healthy tissue homeostasis and the wide range of diseases that involve abnormal tissue mechanics, especially cancer."
The development of an organism is a complex orchestration of cellular processes directed by a genetic blueprint. A range of biochemical cascades work together to create myriad forms and perform many functions that results in life. For years, scientists have focused the biochemistry of embryonic development, Campàs said, because there were not any techniques for measuring mechanical forces that influence cells during the organ and tissue formation.
"We know that the mechanical environment of cells is important," said Campàs. "Growing stem cells on synthetic surfaces with different levels of compliance showed that stem cells would become a different cell type depending solely on the mechanical environment they perceive. If you put embryonic stem cells on a substrate like Jell-O—mechanically similar to brain tissue—they turn into neurons. But if you put them on something harder, similar to embryonic bone, they turn into bone-like cells."
Now, researchers will have a way to investigate the mechanical nature of the cellular environment, where cells are living with other cells inside of live tissue.
"The technique we developed allows the measurement of the mechanical properties such as stiffness and viscosity within living tissues," explained author Friedhelm Serwane, who is currently at the Max Planck Institute for Intelligent Systems in Stuttgart, Germany. "This is exciting because important cell functions are controlled by those mechanical properties. If we can measure the mechanical properties within living organisms now we might be able to understand better how this relationship between mechanics and biology works."
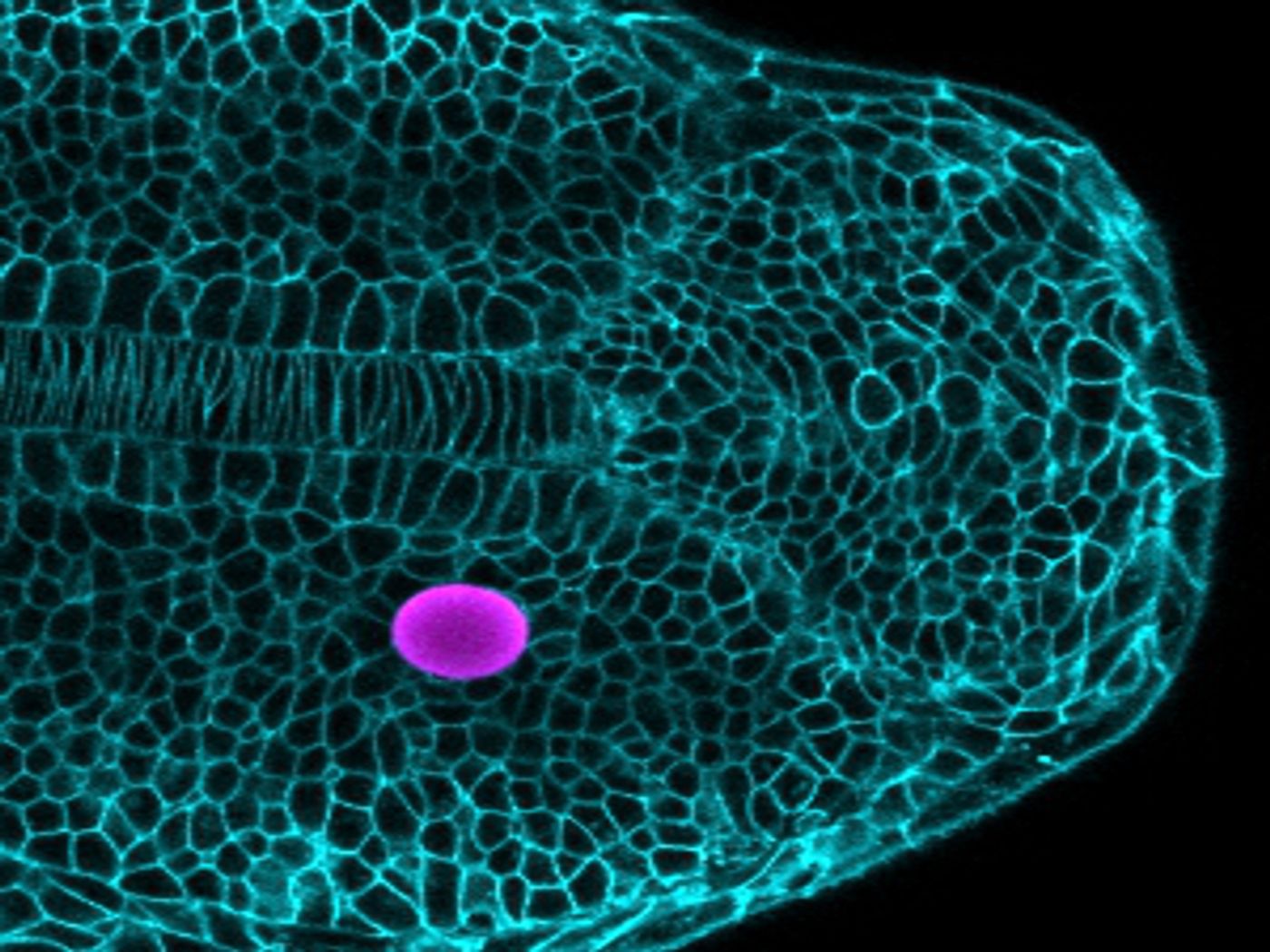
This technique utilizes magnetic droplets put in between cells in the developing embryo. The magnetic droplets deform and push on nearby cells after being exposed to a magnetic field. Careful control of the droplet composition and magnetic field strength enables control of the droplet. The response of the surrounding tissue thus reveals both its mechanical characteristics and the influences on cells as the tissue grows. This technique complements another protocol developed by the Campàs team that revealed the forces that cells have on one another in developing tissues.
The scientists tried their technique on the developing vertebrate body axis of zebrafish, a research organism prized for its fast development and optical transparency. They researchers could show that the tissue’s mechanical characteristics change along the body axis, in a way that facilitates the posterior extension of the body. By inserting magnetic droplets in various locations and applying a magnetic field, the researchers demonstrated that the tissue is fluid as it grows, with thick, dense mechanical properties. The data indicated that the posterior tissue, where growth happens, is more fluid, and it becomes less fluid moving away from the region of growth.
"It is similar to glass-blowing," said Campàs. "The tissue is more fluid in growing regions and 'fixes' its shape by becoming less fluid where it does not need to expand."
These findings could help understand how organs are made, and how cells behave naturally in both healthy and diseased tissues. The Campàs lab is working on a few of these questions, like how limbs are constrcuted and how mechanical changes in tumors affect tumor growth and malignant cell behavior.
Sources: Phys.org via UCSB, Nature Methods