Mathematical Models Reliably Predict Microbial Behavior
There is a huge interest in the development of new antibiotics; they are important drugs and not many new ones come long often. Additionally and more worrisome, bacterial threats are beginning to build and spread resistance to the efficacy of antibiotics, which combat bacteria in a broad way and work by killing all bacteria. However, there are a lot of friendly and beneficial microbes living in our body as well. Researchers have pursued the concept that a newly engineered antibiotic would work in a more tailored, specific way, killing only pathogenic microbes and leaving good ones alone.
"If you could target harmful bacteria in the human gut, you could use engineered bacteria as a living antibiotic," said Brian Hammer, an associate professor at Georgia Tech's School of Biological Sciences. He cautioned, "We're not anywhere near that right now." You can check out a brief TEDx talk Hammer gives in the video to learn more.
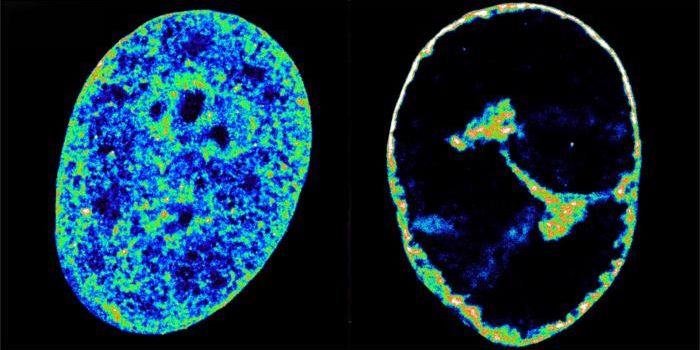
Investigators have to begin by learning more about bacterial behavior over time. To that end, they created mathematical models based on existing physics that characterizes the behavior of molecules in equations. When two strains of cholera were modeled, it was predicted that they would maintain separation, like oil and water. That turned out to be the case in living microbes. "The models predicted pretty much exactly when the phase separation would occur, and then we observed it happening just like the math said it would," Hammer explained.
Vibrio cholera bacteria have a common bacteria weapon in their arsenal - the Type VI secretion system (T6SS); it's a sort of microscopic harpoon that the bacteria use at random, stabbing whatever bacteria comes close. Other cholera bacteria are immune to the effects of the harpoons. All kinds of bacteria use the T6SS system.
"This harpoon system is in about one quarter of Gram-negative bacteria," Hammer said. "So, this bacterial dueling is going on all around you." Bacteria that win these competitions are generally better are surviving; they work together to create a cooperative community and importantly, to share food. That food store has to be managed, however.
"Bacteria do a lot of their digestion outside their cells. You need a strategy for ensuring that all the effort of chewing up and digesting food benefits you and your relatives, and not someone else who comes and plunders it," explained Hammer.
When a bacterial strain is able to kill invaders, it also passes on its own genes as it grows and creates future generations. The investigators assayed the genomes of various bacteria with poison harpoon systems, some having multiple poisons or harpoons. "We found that the more weaponry a bacteria strain had in its genome, the more it looked like it was apt to share," Hammer revealed.
The researchers used bacteria stained either red or blue, which were then visualized in a microscope as they harpooned it out. "We start with two strains well mixed," Hammer said. "We jokingly call this the salad dressing model, because you shake oil and water, and they're well mixed, and you let it sit, and they phase separate."
A purple mass became separate blotches of red and blue after territory is staked out and battles are won by the various strains. The researchers found that despite the differences, mathematical equations predicting chemical behavior also worked for microbes.
"The type of curve we observed describing our results had never been used to describe living systems before," Hammer said. "Empirically, it's been used to describe metals that undergo phase separation."
The researchers have published their findings, including accurate models, in Nature Communications. "The mathematical simulations overlapped with the observed data almost perfectly," Hammer said.
These findings could aid future clinical therapeutics. "In your gut, a lot of useful bacteria are Gram-positive," Hammer said. "But there might be a small number of Gram-negative bacteria messing up your gut community, and perhaps engineered bacteria with spears could get rid of just those Gram-negative."
Sources: AAAS/Eurekalert! via Georgia Tech, Nature Communications