Using Stem Cells, Scientists Generate an Artificial Mouse Embryo
Researchers have used two types of stem cells and a three dimensional scaffold to generate a structure like a mouse embryo. The researchers are hoping this work could provide insight into why many pregnancies fail. When the artificial embryo was compared to a real mouse embryo, the scientists found that it was on the same developmental course as mouse embryos. The work is published in Science and summarized in the following video.
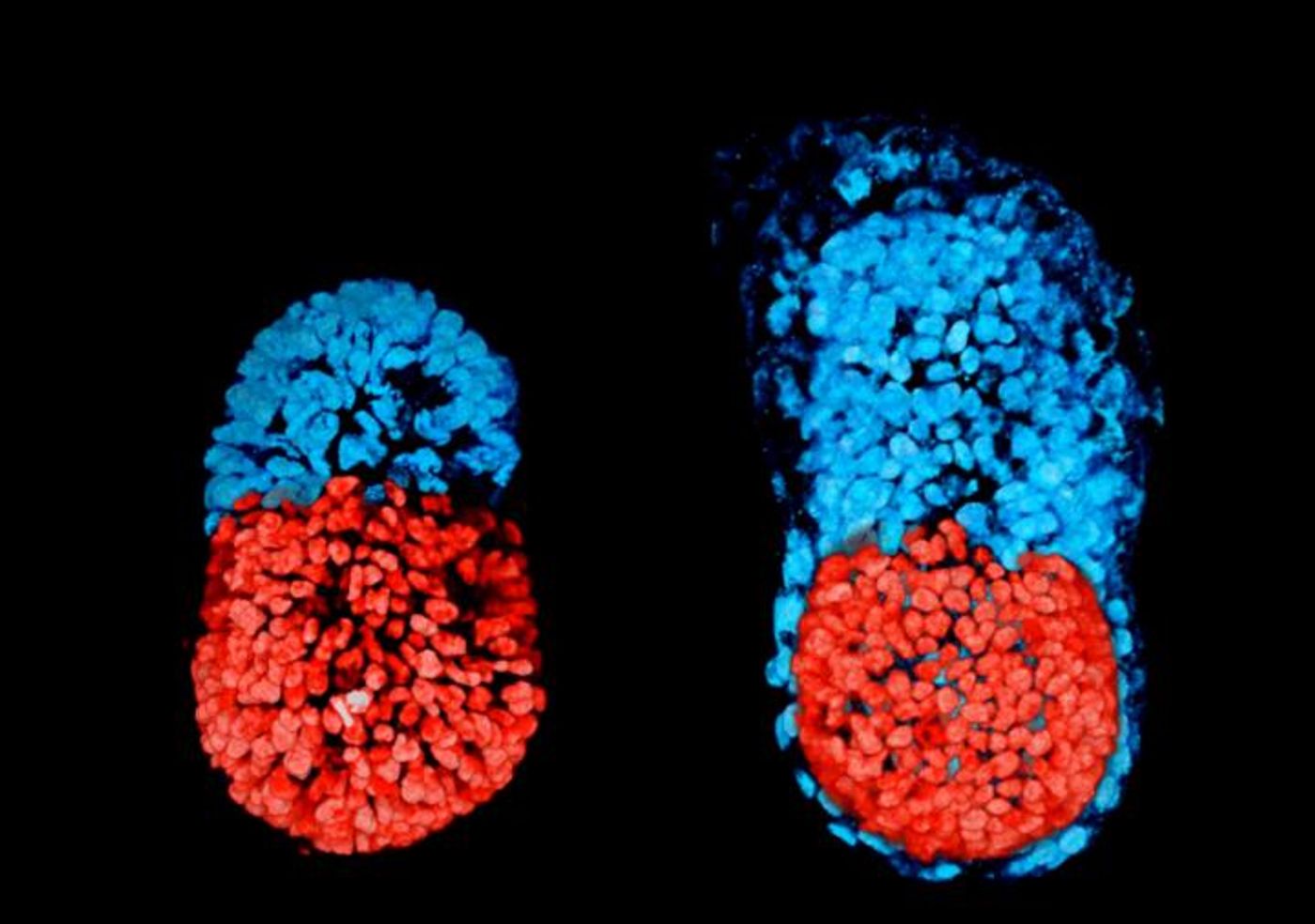
When a sperm fertilizes a mammalian egg, cell division begins and a ball of stem cells is subsequently generated. An organism grows from embryonic stem cells (ESCs) that aggregate near one end of the developing embryo or blastocyst. At the other end are trophoblast stem cells (TSCs), cells outside the embryo that that give rise to the placenta and primitive endoderm cells, from which the yolk-sac is generated to ensure nutrition and proper organ development.
While there have been other attempts to grow structures resembling embryos, there has not been much success to this point. It appears that different kinds of cells are an essential requirement, from that cooperative effort an embryo-like structure was created. "Both the embryonic and extra-embryonic cells start to talk to each other and become organized into a structure that looks like and behaves like an embryo," said the leader of the work, Professor Magdalena Zernicka-Goetz of the Department of Physiology, Development and Neuroscience. "It has anatomically correct regions that develop in the right place and at the right time."
The research team observed a significant amount of communication between these different kinds of stem cells; the cells were working together to orient each other properly in the embryo. "We knew that interactions between the different types of stem cell are important for development, but the striking thing that our new work illustrates is that this is a real partnership - these cells truly guide each other. Without this partnership, the correct development of shape and form and the timely activity of key biological mechanisms doesn't take place properly," she explained.
The structure formed by the organized cells, with ESCs and TSCs going to opposite ends. A cavity opens within each group and then the merges. Eventually the large, pro-amniotic cavity is created and it is there the embryo develops. In this case however, the researchers stress that it's unlikely that this artificial embryo would continue to develop into a fetus. The artificial embryo is missing a critical third layer of cells that enables yolk sac development and allows blood vessels to form. The placenta would also be unable to grow without additional optimization.
Zernicka-Goetzhas also pioneered a method enabling blastocysts to grow beyond the implantation stage, in vitro, up to 13 days after fertilization; researchers are thus able to closely analyze very early stages of human development for the first time. She suggested that the newest work can help provide laboratories with research models that were previously unavailable, and opens up the door to research on human embryos, without having to obtain them from donors.
"We think that it will be possible to mimic a lot of the developmental events occurring before 14 days using human embryonic and extra-embryonic stem cells using a similar approach to our technique using mouse stem cells," she said. "We are very optimistic that this will allow us to study key events of this critical stage of human development without actually having to work on embryos. Knowing how development normally occurs will allow us to understand why it so often goes wrong."
"This is an elegant study creating a mouse embryo in culture that gives us a glimpse into the very earliest stages of mammalian development. Professor Zernicka-Goetz's work really shows the importance of basic research in helping us to solve difficult problems for which we don't have enough evidence for yet. In theory, similar approaches could one day be used to explore early human development, shedding light on the role of the maternal environment in birth defects and health," commented Dr Andrew Chisholm, the Head of Cellular and Developmental Science at Wellcome.
Sources: AAAS/Eurekalert! via University of Cambridge, Science