A Health-promoting gut Microbe Adapted to Human Breast Milk
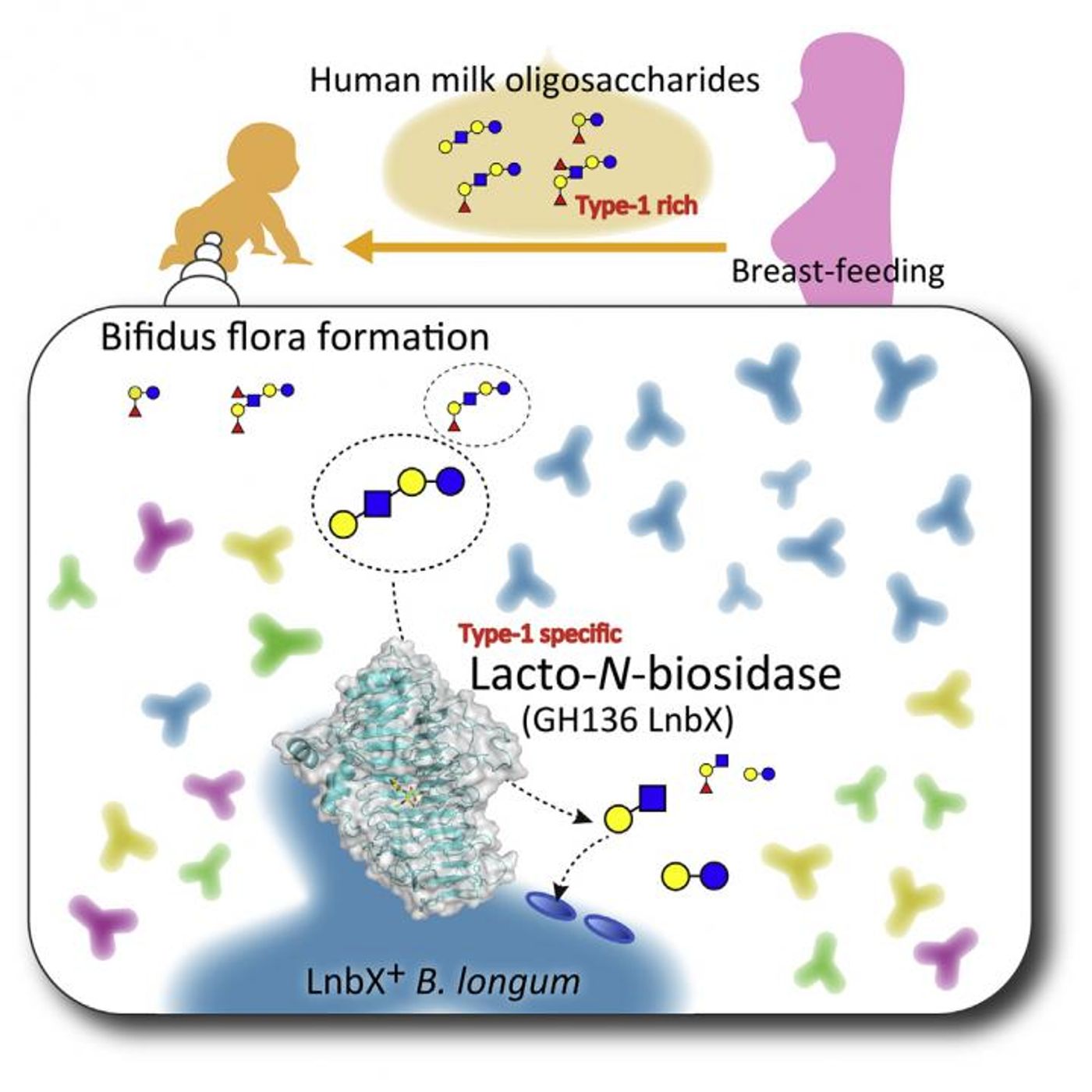
Breast milk is known to supply infants with important nutrition and immunity; new work indicates it also supports the health of beneficial microbes that reside in the gastrointestinal tract, or human gut microbiome. The work, reported in Cell Chemical Biology, demonstrates that a bacterial species called Bifidobacterium longum has adapted specifically for growing in the infant gut; it is able to produce an enzyme that allows it to utilize a sugar in human milk for energy.
"Given the health-promoting effects of bifidobacteria, our findings reveal a possible evolutionary route for the breast milk-driven symbiosis between gut microbes and humans," said co-senior author of the report, Takane Katayama of Kyoto University.
Evidence continues to mount that the microbiome has a big influence on human health, and it can start on the road to good health at or even before birth. Diet can have a big impact on the makeup of the microbiome. It is known that human milk sugars can promote the growth of beneficial gut microbes such as bifidobacteria specifically. That bacterium can prevent diarrhea and infection in infants. A sugar called lacto-N-tetraose is present at high levels in human milk while it’s not really found in the milk of other mammals. Bifidobacteria synthsize enzymes that can metabolize this sugar, suggesting the evolution of a symbiotic relationship.
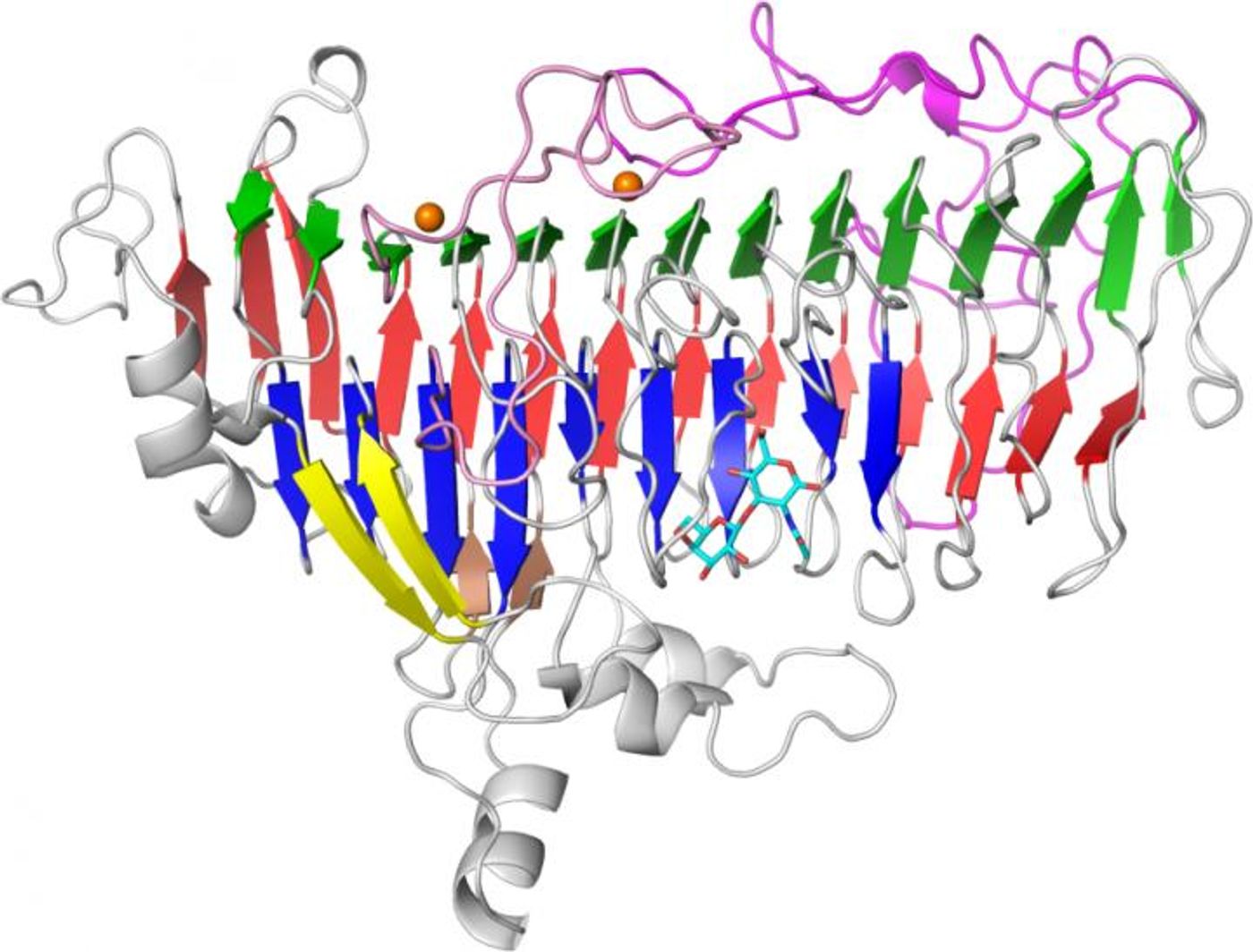
To investigate the evolution of that relationship further, Katayama and co-senior study author Shinya Fushinobu of the University of Tokyo have characterized the LnbB enzyme and isolated the LnbX enzyme, which break down lacto-N-tetraose in Bifidobacterium bifidum and Bifidobacterium longum, respectively. Building on that previous work, the scientists identified the X-ray crystal structure of LnbX’s catalytic domain. Taken with other data, it was found that the structure and catalytic mechanism of LnbX are different from LnbB, thereby placing it in a new family of glycoside hydrolase enzymes termed GH136.
"Even though B. longum and B. bifidum belong to the same genus and inhabit the same environment, they use different enzymes to break down lacto-N-tetraose, taking advantage of the varied structures of this unique human milk sugar," Fushinobu explained. "The findings suggest that different strains and species of beneficial bifidobacteria have independently evolved distinct molecular tools to digest the same human milk sugar, explaining their ability to co-exist and thrive in the gastrointestinal tract of breast milk-fed infants."
The investigators also found that B. longum requires lnbX gene expression for it to be able to grow specifically with lacto-N-tetraose. Fecal DNA analysis showed B. longum and the lnbX gene are present at higher levels in the microbiome of ten infants that were only fed breast milk compared with six infants that were given a mixture of formula and breast milk. "Taken together, these findings suggest that lnbX is important for B. longum to persist in the gut ecosystem of breast milk-fed infants, and human milk sugars have been the main selective pressure for the evolution of lnbX," Katayama explained.
The researchers are continuing this work; they want to determine if other bifidobacterial species make other enzymes to metabolize breast milk sugars. They also plan to find bifidobacterial metabolites that are beneficial to infant health with a goal of improving formula milk. That could be by adding beneficial bacterial compounds or enzymes.
"Although breast feeding during the first year of life is recommended in most cases, some mothers have to rely on formula milk because they have viral infections or do not produce milk with key nutrients such as zinc," Katayama said. "Therefore, the development of new strategies to fortify formula milk with health-promoting ingredients will be especially critical in these rare cases where breast milk feeding is not possible."
You can learn more about the benefits of breastfeeding in the video.
Sources: AAAS/Eurkealert! via Cell Press, Cell Chemical Biology