New, Powerful Antibiotic Found in Soil
Soil has previously been the source of effective antibiotics, and now it has produced another. A new antibiotic called pseudouridimycin is made by a microbe that was collected in a soil sample taken in Italy. It effectively destroyed a broad spectrum of bacteria that are drug-sensitive and -resistant when assayed in vitro, and was able to cure a bacterial infection in a mouse model.
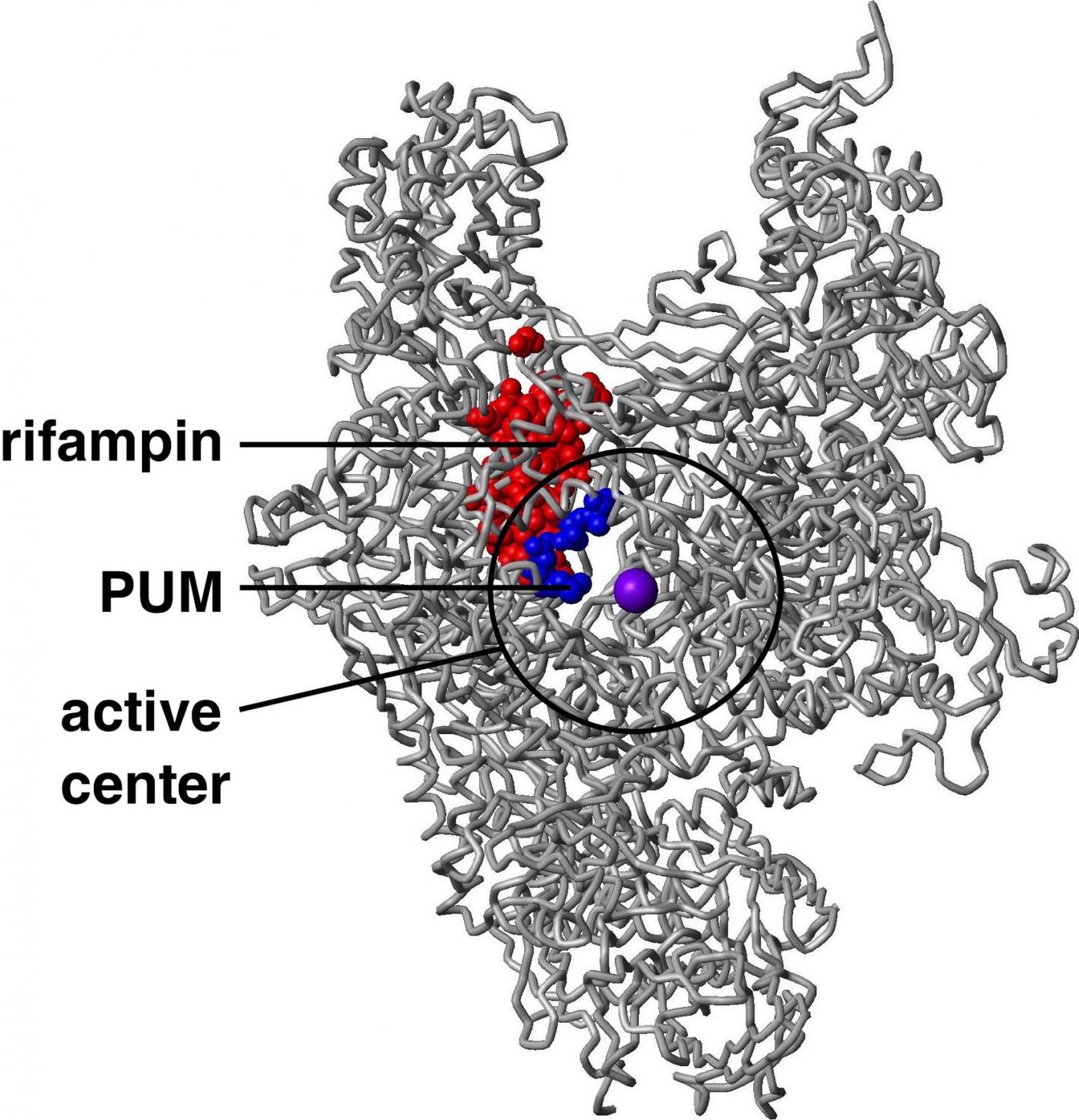
Researchers at Rutgers University-New Brunswick, the biotechnology company NAICONS Srl., and elsewhere reported the discovery in Cell, along with the mechanism behind the antibiotic’s action. The pseudouridimycin acts by inhibiting an enzyme, bacterial RNA polymerase, which makes RNA molecules in bacteria, essential for their survival.
Related: Antibiotics, Antibiotic-resistance Comes From the Same Place
Pseudouridimycin can mimic one of the ‘bases’ that RNA polymerase uses to build RNA molecules, nucleoside-triphosphate (NTP). As such it is a nucleoside-analog inhibitor of the polymerase. The drug attaches to the binding site used by NTP on the polymerase and stops new NTPs from binding there, stopping synthesis of the essential new RNA. Interestingly, this is the first nucleoside-analog inhibitor that is effective against bacterial RNA polymerase while leaving human RNA polymerase alone.
"Because the NTP binding site of bacterial RNA polymerase has almost exactly the same structure and sequence as the NTP binding sites of human RNA polymerases, most researchers thought it would be impossible for a nucleoside-analog inhibitor to inhibit bacterial RNA polymerase but not human RNA polymerases," said Richard H. Ebright, Board of Governors professor of chemistry and chemical biology and laboratory director at the Waksman Institute of Microbiology at Rutgers-New Brunswick, who led the research.
"But pseudouridimycin contains a side-chain that 'reaches' outside the NTP binding site and 'touches' an adjacent site that is present in bacterial RNA polymerase but not in human RNA polymerases and, as a result, it binds more tightly to bacterial RNA polymerase than to human RNA polymerases," Ebright said.
The antibiotic is an alternative to another commonly used drug that inhibits the same enzyme, a drug called rifampin. Since its mode of action is different from rifampin pseudouridimycin can be used in conjunction with it for a powerful, additive effect. Additionally, it has no cross-resistance with rifampin. Perhaps most critically, it has one-tenth the spontaneous resistance rate of rifampin.
The low rate of resistance is probably attributable to the fact that pseudouridimycin is a nucleoside-analog inhibitor. "The new antibiotic interacts with essential residues of the NTP binding site that cannot be altered without losing RNA polymerase activity and bacterial viability," Ebright explained. "Alterations of the NTP binding site that disrupt binding of the new antibiotic also disrupt RNA polymerase activity, resulting in dead bacteria, rather than resistant bacteria."
"Nucleoside-analog inhibitors that selectively inhibit viral nucleotide polymerases have had transformative impact on the treatment of HIV-AIDS and hepatitis C," said research co-leader Stefano Donadio, CEO of NAICONS Srl. "The anti-AIDS drugs Zidovudine, Videx, Zalcitabine, Lamivudine, and Viread are nucleoside-analog inhibitors, and the anti hepatitis-C drugs Solvadi and Harvoni are nucleoside-analog inhibitors."
"Nucleoside-analog inhibitors that selectively inhibit bacterial RNA polymerase could have a similarly transformative impact on the treatment of bacterial infections," Donadio continued.
"The discovery also underscores the importance of natural products in providing new antibiotics. Microbes have had had billions of years to develop 'chemical weapons' to kill other microbes,” he concluded.
Learn more about how nucleoside-analog inhibitors work form the video above, which uses AIDS drugs as an example.
Sources: AAAS/Eurekalert! via Rutgers University, Cell