Therapeutic antibodies for Alzheimer's disease: challenges and hopes
Solanezumab: this simple name evokes at the same time one of the greatest hopes and the worst disillusions for Alzheimer’s Disease (AD) patients. Two years ago, this very expected monoclonal antibody (by Eli Lilly) supposed to slow down - or even stop - AD progression finally failed in the third and last stage of its clinical trials. Despite its successful results in animal models, Solanezumab could not fulfill its promises on humans as it did not bring any significant improvement in AD patients’ cognitive functions nor survival. This experience has nonetheless greatly contributed to the development of passive immunization and new hopes have risen. Here is an overview of the improvements to come.
Identifying the main issues
More than a century since the first case of Alzheimer was described, its process remains a mystery. This unstoppable degenerative disease, characterized by progressive dementia, affects more and more people every year. The number of AD patients could reach 130 million in 2050, making it one of the significant unmet medical needs in modern medical history. Existing therapies are only limited to symptomatic relief, but many studies are now focusing on curative treatments.
Two main hypotheses are currently guiding most of the therapeutic researches.
The AD is associated with various cellular dysfunctions, including the development of senile plaques composed of the amyloid β peptide (Aβ). According to the “amyloidbeta cascade hypothesis,” Aβ aggregation leading to the formation of these senile plaques is the primary upstream event in AD pathogenesis.
An intraneuronal accumulation of neurofibrillary tangles (NFTs) is also observed in AD patients. The Tau hypothesis is based on the assumption that their development is the driving pathogenic element explaining the neuronal loss.
It is also not to be excluded that the Aβ plaques and the neurofibrillary tangles might both result from an unknown upstream factor. Other troubles are also observed in AD, such as cholinergic neuron damages and oxidative stress, and inflammatory reactions.
All these pathologic expressions are obviously linked to each other, but it is rather complicated to determine the chicken and egg within the global AD process.
Improving the existing treatments
The majority of the recent researches are focusing on preventing the aggregation or stimulating the clearance of Aβ plaques. One of the most promising methods so far relies on the emergence of immunotherapy techniques. Although no curative treatment has reached the market yet, preclinical studies have shown the best-known results in animal models.
The active immunization aims to stimulate the inner immune system against the pathological expressions of the disease. It acts as a vaccine: fragments of Aβ antigen are administered in order to induce an immune response. Unfortunately, this technique generated unacceptable rates of severe side effects during the clinical tests, such as meningoencephalitis.
In response, scientists developed passive immunization as a safer alternative, which showed great success in mice for the clearance of Aβ plaques. This technique consists in the direct administration of monoclonal antibodies, designed to address the formation or the accumulation of Aβ.
Unfortunately, the recent trials of Bapineuzumab, Ponezumab, and Solanezumab could not bring to any safe improvement of survival and cognitive functions. In light of these failures, several sources of improvement are explored aiming to replicate the promising results obtained in preclinical studies.
Increasing the antibodies uptake by improving their access to the brain
In previous AD immunotherapy studies, it appeared that a very poor rate (approximately 0,1%) of the injected monoclonal antibodies was able to reach the brain. This is attributable to the existence of the blood-brain barrier (BBB), aiming to prevent the access of most proteins from the blood to the CNS.
A possible strategy to outpace this filter could rely on bispecific antibodies able to bind to both a BBB receptor and the antigen of interest. The use of focus ultrasound (FUS) is also considered for this purpose. This technique generates microbubbles that allow the penetration of biotherapeutics into the CNS through the BBB.
Intervening more efficiently thanks to an earlier diagnosis
As Aβ aggregation usually starts more than a decade before the first AD symptoms, another way of improving the effectiveness of the treatments would be to administrate them at an early, asymptomatic stage of the disease.
A few years ago, the detection of amyloid plaques required either biopsy or postmortem histopathology. Nowadays, new imaging technologies such as PET-scan linked with amyloid-plaques labeling ligands allow their detection in living patients, but their presence remains the sign of an advanced stage of the disease. This factor may explain the inefficacy of some Aβ targeting treatments, such as Solanezumab. Improving the diagnosis by developing new biomarkers and methods in order to detect abnormal Aβ conformations earlier is, therefore, a major issue for pharmaceutical industries.
Preventing the development of side effects
Bapineuzumab trials were aborted for provoking cases of vasogenic edema and microhemorrhages. These complications resulted from the activation of antibody’s effector function. To limit the microglial activation leading to inflammatory responses, a modified Fc receptor was tested in AAB-003, a Bapineuzumab alternative. This attempt was finally aborted for no bringing significant improvements but could be enhanced in future therapeutics. A slow and progressive increase in the dose administrated is another option that could help to prevent some important side effects.
Exploring new paths
Facing the failure of Aβ therapies, new directions are progressively considered.
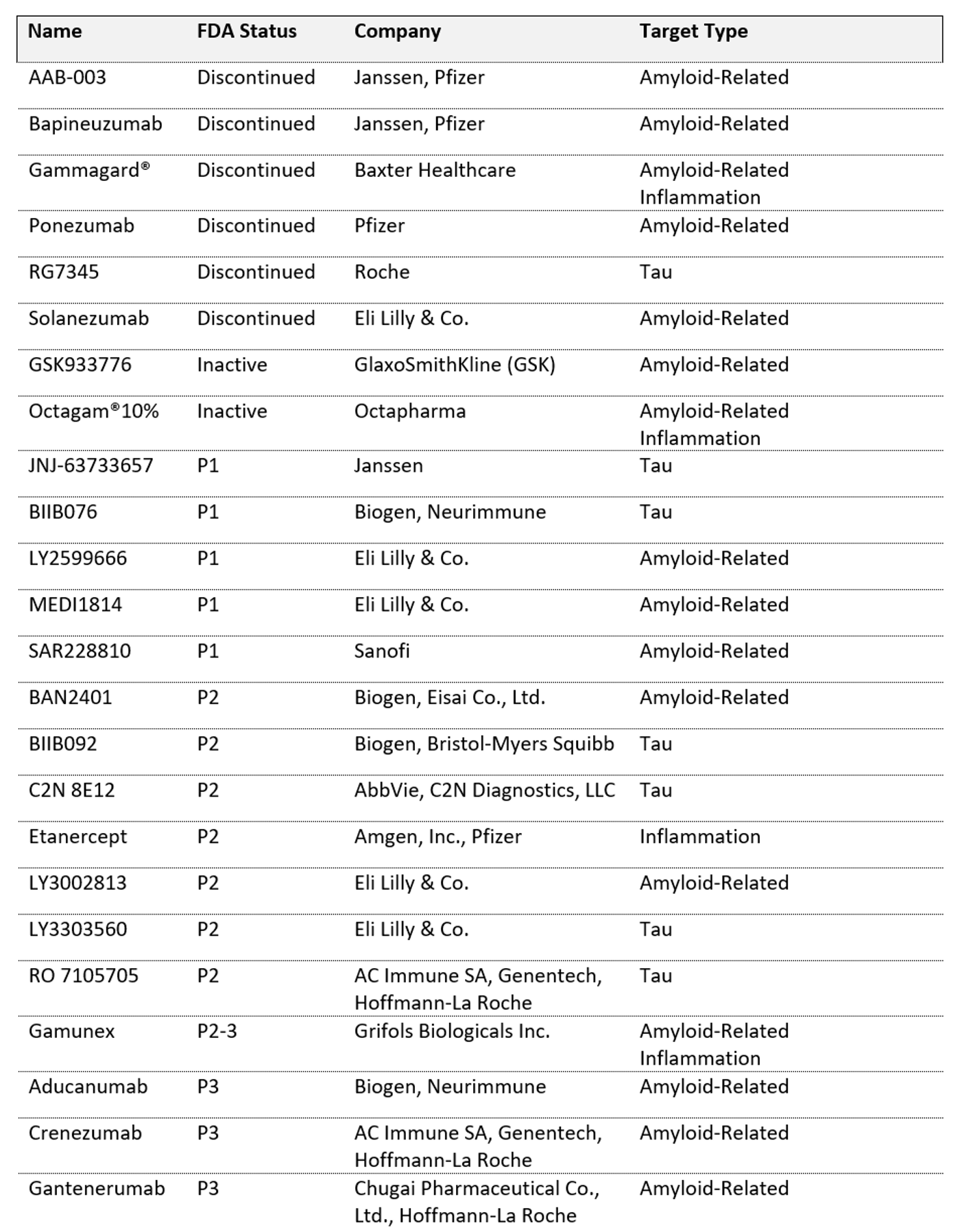
Tau hypothesis seems to take more and more credit, as reflected by the increasing number of Tau fibrils-targeted antibodies emerging in early clinical phases. This new type of targets concerns 6 of the 12 therapeutic antibodies currently tested on first and second phases of clinical trials (see table below).
Furthermore, a common point between Aβ and Tau pathology is the abnormal oligomer forms associated. Incoming studies are focusing on antibodies targeting these conformers. They may allow acting on both pathologies at the same time.
Other paths relying on monoclonal antibody production are also explored, such as the cholinergic and oxidative stress hypothesis and the inflammatory hypothesis.
Overview of passive immunotherapy trials for Alzheimer’s disease (source: alzforum.org, 2018)
The future success of immunotherapy in AD may rely on several advances in imaging and biotechnologies. The use of monoclonal antibody engineered products resulting from antibody fragments, Antibody Drug Conjugates (ADC), bifunctional or bispecific antibody development is seriously considered. Using these techniques may be a key to bring major advances and hopes to AD patients in a near future.
Sources
Aisen, P.S., and Vellas, B. (2013). Passive immunotherapy for Alzheimer’s disease: What have we learned, and where are we headed? J. Nutr. Health Aging 17, 49–50.
Beck, A., Wurch, T., Bailly, C., and Corvaia, N. (2010). Strategies and challenges for the next generation of therapeutic antibodies. Nat. Rev. Immunol. 10, 345–352.
Cappa, S.F. (2018). The Quest for an Alzheimer Therapy. Front. Neurol. 9.
Du, X., Wang, X., and Geng, M. (2018). Alzheimer’s disease hypothesis and related therapies. Transl. Neurodegener. 7.
Kametani, F., and Hasegawa, M. (2018). Reconsideration of Amyloid Hypothesis and Tau Hypothesis in Alzheimer’s Disease. Front. Neurosci. 12.
Schilling, S., Rahfeld, J.-U., Lues, I., and Lemere, C. (2018). Passive Aβ Immunotherapy: Current Achievements and Future Perspectives. Molecules 23, 1068.
Spencer, B., and Masliah, E. (2014). Immunotherapy for Alzheimer’s disease: past, present and future. Front. Aging Neurosci. 6.
Tarawneh, R., and Holtzman, D. (2009). Critical Issues for Successful Immunotherapy in Alzheimer’s Disease: Development of Biomarkers and Methods for Early Detection and Intervention. CNS Neurol. Disord. - Drug Targets 8, 144–159.
Wisniewski, T., and Goñi, F. (2014). Immunotherapy for Alzheimer’s disease. Biochem. Pharmacol. 88, 499–507.
This article was written by Sophie Le Ray, a scientific writer and communication officer for ProteoGenix, a leading custom assay and therapeutic antibody development service provider. We wish to share our expertise with the scientific community by providing quality content about the latest and most promising advances in biotechnology.