How does the brain know when we are full?
Thanksgiving is around the corner. All that amazing food. One more slice of that amazing pie, but our brain says “Full! Stop Eating”. How does the brain know we are full?
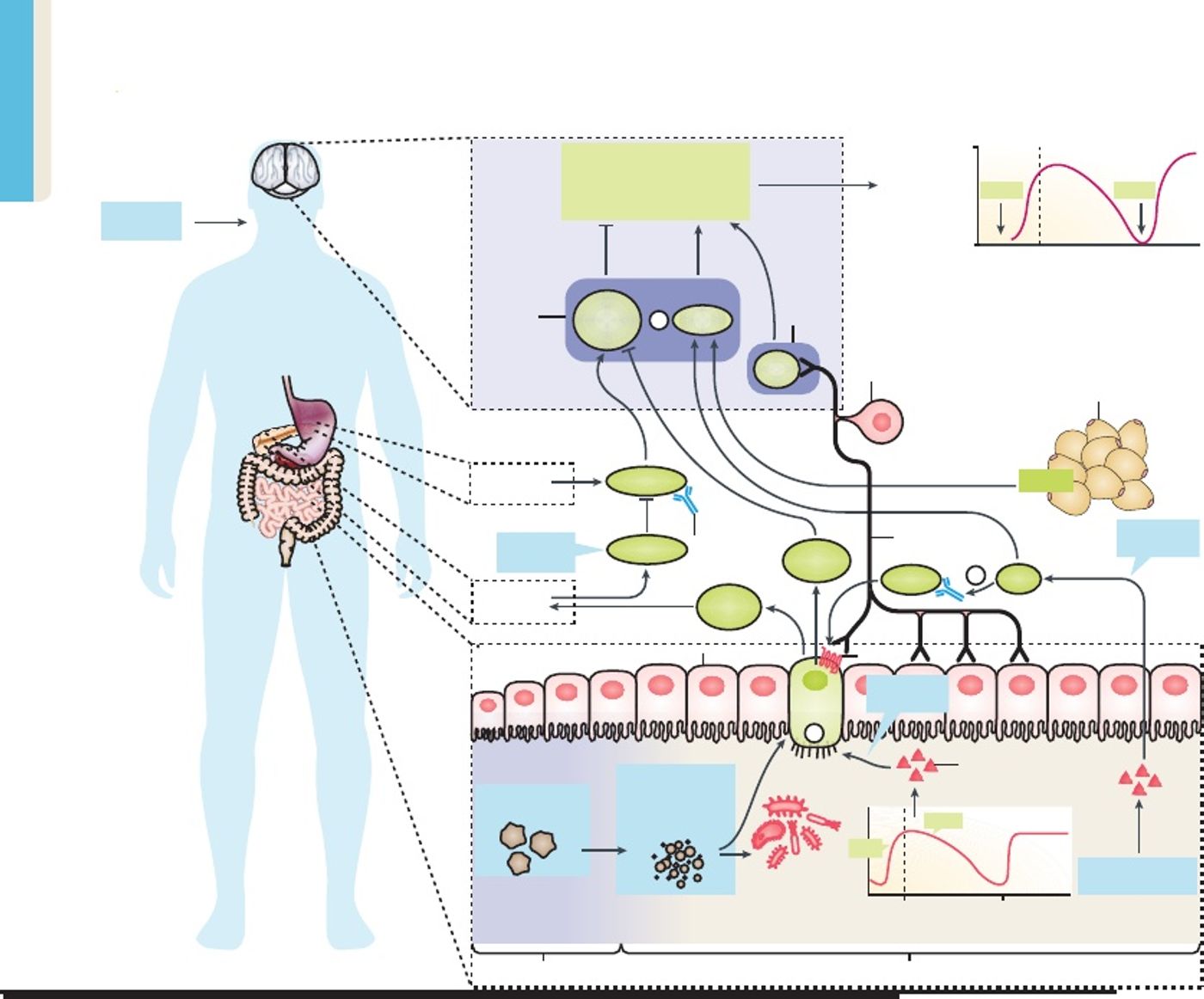
Feeling full or satiation is conveyed to the brain by the gut hormones via the enteric neuronal afferents and the endocrine feedback pathways. Serguei Fetisov, Professor of Physiology at Rouen University, France says "Gut bacteria physiologically participate in appetite regulation immediately after nutrient provision by multiplying and stimulating the release of satiety hormones from the gut. Also, we believe gut microbiota produce proteins that can be present in the blood longer term and modulate pathways in the brain." Dr. Fetisov has extensively studied the role of gut microbiota in appetite control. Although models have been proposed to understand the complex molecular pathways involved this communication process, there has been no evidence so far.
Bacteria–host integrative homeostatic model of appetite control. Image Credit: Fetisov et al., Nature Reviews 2016
A study published in Cell Press on November 15th, by researchers from Germany and Finland have shown that brown-fat interacts with gut hormone secretin to relay appetite control signals to the brain. Brown adipose tissue (BAT) is a type of body fat that is primarily involved in thermoregulation and produces non-shivering thermogenesis when the mammal is cold.
“We demonstrate a connection between the gut, the brain, and brown tissue, uncovering a previously unknown facet of the complex regulatory system controlling energy balance,” says lead author Martin Klingenspor, chair of molecular nutritional medicine at the Technical University of Munich. “The view of brown fat as a mere heater organ must be revised, and more attention needs to be directed towards its function in the control of hunger and satiation.”
The authors hypothesized that one or more of the gut hormones could be associated with activation of BAT and thereby activate meal associated non-shivering thermogenesis. Previously, secretin, a gut hormone was considered to encode the meal information and plays a role in the central perception of satiation. Gene expression of the BAT tissue in mice showed abundant expression of secretin and adipocyte cultures provided evidence that secretin activated thermogenesis. Expanding these observations to appetite control situations showed that hungry mice, when injected with secretin, had suppressed appetites with increased BAT induced thermogenesis. The author’s say that while secretin induced thermogenesis in BAT accelerates satiation, the absence of secretin decreases the thermogenesis, increase food intake and eventually changes the meal patterning.
The researchers also investigated the secretin-BAT interaction in humans. Secretin levels, BAT oxygen consumption and fatty-acid uptake measured in 17 healthy human volunteers after an overnight fast and 30-40 minutes after a meal showed that higher the level of secretin in the subjects blood corresponds to higher metabolic activity in the BAT.
Klingenspor says that one day, we may know enough about the secretin-brown fat connection to stimulate secretin production by eating certain foods. “Any stimulus that activates brown fat thermogenesis could potentially induce satiation,” he says. “Secretin secretion is sensitive to nutrients, so eating the right starter could be helpful in promoting satiation and result in reduced meal size and caloric intake.”
He believes that brown fat’s roles in controlling hunger and satiation make it a particularly attractive target for new approaches to treating obesity. Targeting brown fat through secretin might hold promise for potential future nutritional or pharmacological interventions against obesity and metabolic disease, he says.
The TED-Ed Animation lesson is an excellent source for more details on how these signals are transcribed to the brain.
Sources: Cell Press, Neuroscience News