Researchers identify neural pathways that control behavioral responses to noxious stimuli
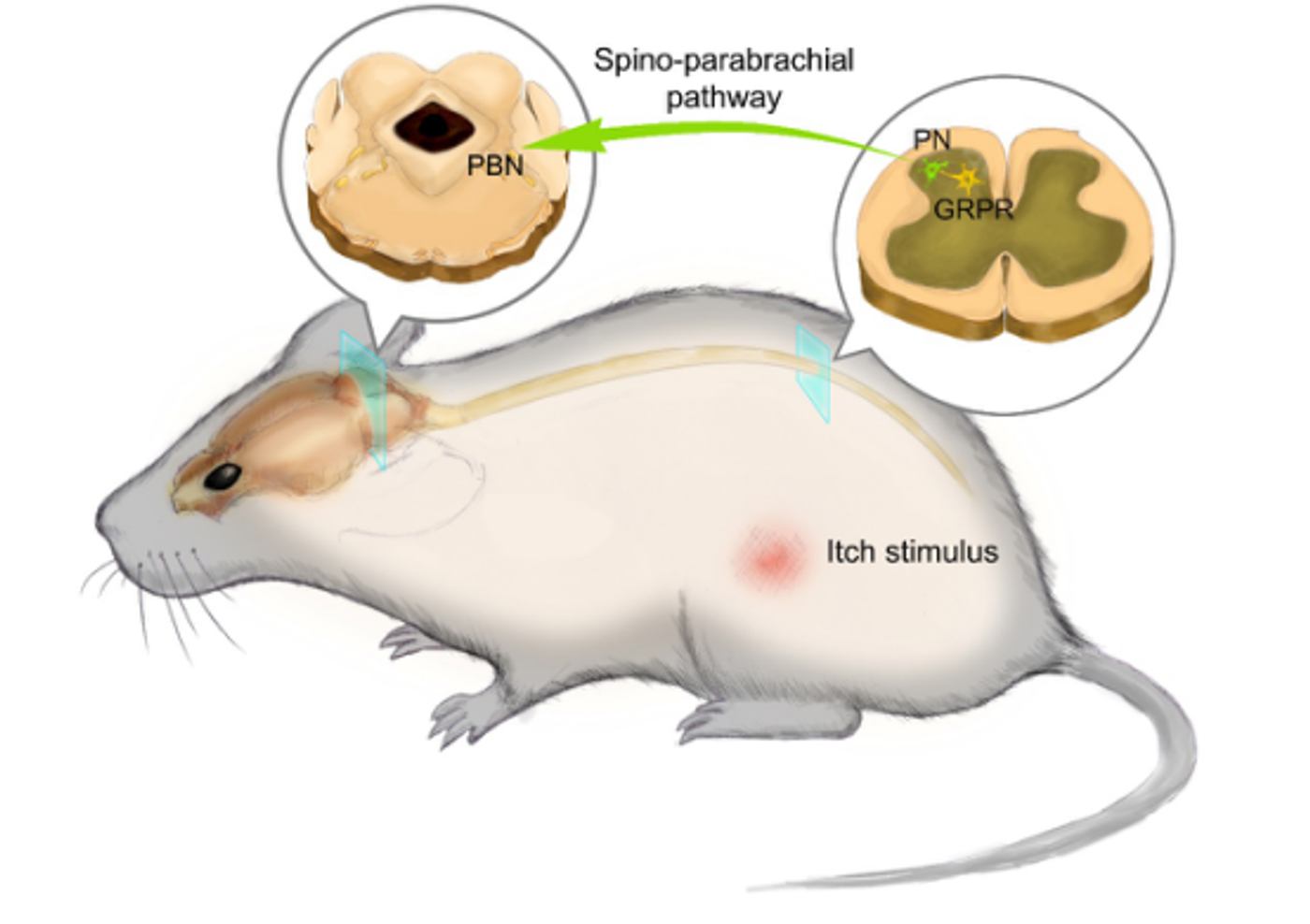
Behavioral responses to the pain could range from the reflexive withdrawal to more complex behaviors to avoid or decrease the pain. Neurons in the lateral division of the parabrachial nucleus (PBNI) are deemed to be the primary responders to various noxious stimuli. However, information on the subtypes of neurons within the PBNI that respond to pain have not yet been explored.
View this video that animates the five phases of nociceptive pain.
Decerbrated preclinical animal studies showed that supra-spinal hindbrain circuits also play a critical role in pain perception. As with the PBNI, neuronal substrates within this circuit, that individually respond to noxious stimuli remain unknown.
Image Credit: SUN Yangang's lab, University of Chinese Academy of Sciences, China
Study published in Neuron by researchers from the National Center for Complementary and Integrative Health (NCCIH), an intramural division of NIH, identified a small group of Tachykinin(Tac-1) positive neurons in PBNI, that receive monosynaptic input from the dorsal spinal cord and make excitatory connections with the dorsal reticular formation in the medulla (MdD) in response to noxious heat stimuli.
“Much is known about local spinal cord circuits for simple reflexive responses, but the mechanisms underlying more complex behaviors remain poorly understood,” said Alexander T. Chesler, Ph.D., a Stadtman Investigator at NCCIH and senior author of the study. “We set out to describe the brain pathway that controls motor responses and involuntary behaviors when the body is faced with painful experiences.”
The gene Tac1 encodes tachykinin peptide hormones, which are known to induce behavioral responses and function as vasodilators and secretagogues. Racing of the neurons within PBNI showed their projections to MdD. Further chemogenetic activation of PBNI neurons using DREADD in 6 mice showed that when presented with noxious heat stimulus, escape like jumping behavior was seen. This response was absent when the heat below pain threshold was introduced confirming the role of PBNI neurons in painful somatosensory signaling.
Further, ablation of the PBNI Tac1 neurons showed no jumping behavior in response to a painful heat stimulus. Tracing of the PBNI Tac1 neurons also revealed that they receive input from two primary anterograde targets, MdD and central nucleus of the amygdala (CeA) with MdD as the most crucial target. Additionally, a subset of Tac1 expression neurons was also found in MdD with protections to dorsal spinal cord and PBN. Together, PBNI and MdD Tac1 neurons create a pronociceptive Tac1 brainstem-spinal circuit that regulates the behavioral responses to noxious stimulus.
“Our data provide evidence that the PBNI produces streams of information with distinct functional significance,” said Arnab Barik, Ph.D., a postdoctoral fellow at NCCIH and one of the study’s authors. “The brainstem-spinal cord pathway identified in this study selectively controls pain response and elicits appropriate behaviors based on sensory input.”
Added Chesler, “This kind of feed-forward circuitry is unique because it is an upward spiral. The more this pathway is activated by harmful activity, the more it reacts, leading to dramatic behavioral responses.”
These findings are just the tip of the iceberg. Tac1 expressing neurons might be the gatekeepers but how this information trickles down to complex behavioral responses in the presence of other biological demands warrants more research. Understanding these complex pain pathways might help us understand how homeostasis is affected during chronic pain.
Sources: Neuron, Neuroscience News