The Antidepressant in Everyone's Medicine Cabinet: The case for the inflammatory theory of depression grows
Compared to the countless number of serotonin and dopamine-centered depression studies, the kynurenine pathway (KP) holds less research attention. However, with recent findings essentially dethroning the monoamine theory of depression, kynurenine might soon have its day in the funding sun.
To be fair, it’s possible that numerous anti-depression therapies work through shared pathways, including KP. For example, serotonin has displayed anti-inflammatory properties. However, studies and reviews can’t seem to agree if inflammation increases or decreases its concentrations.
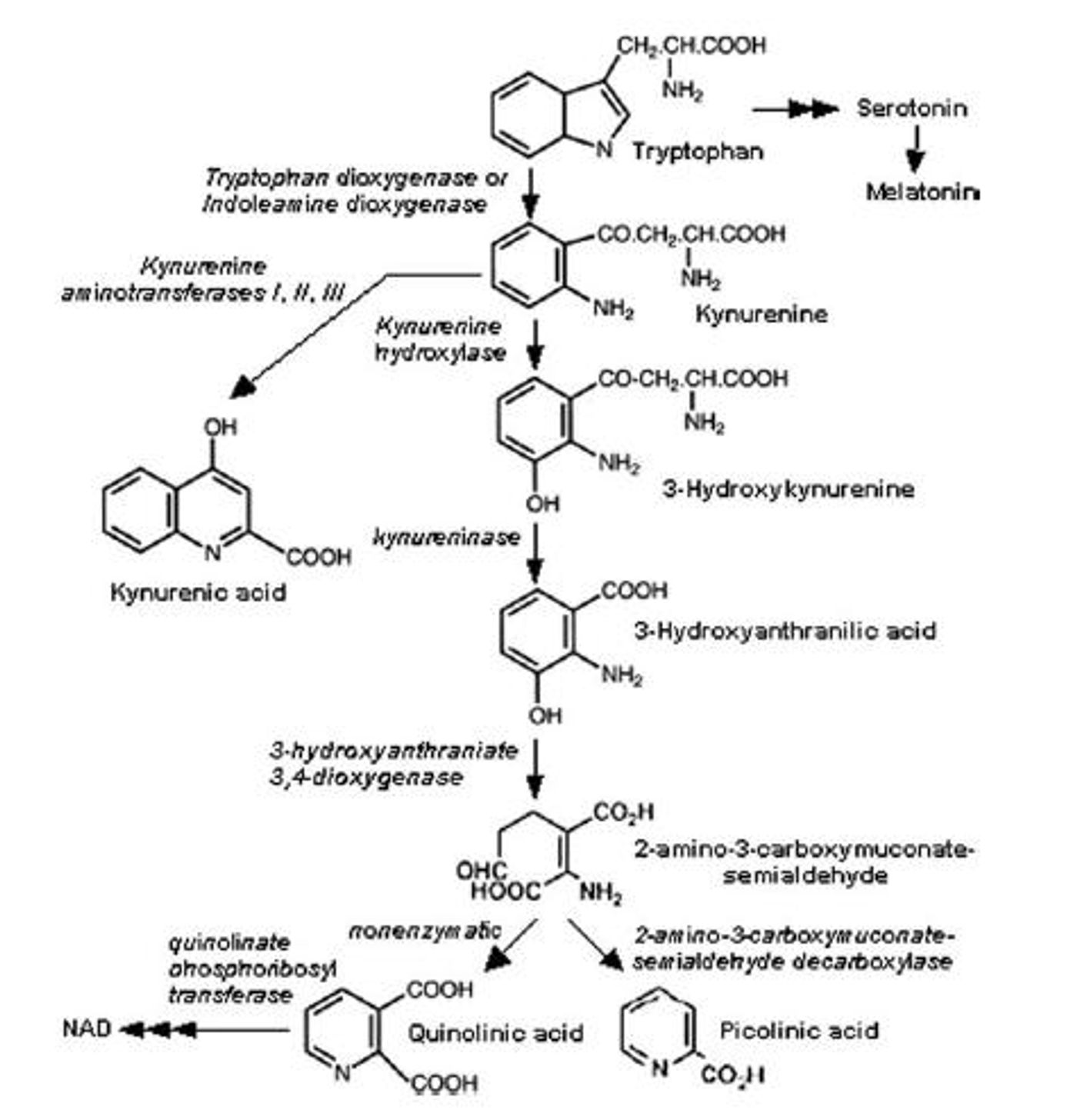
The KP is an alternative direction for metabolizing tryptophan as opposed to synthesis into serotonin. New research suggests nonsteroidal anti-inflammatory drugs (NSAID), such as ibuprofen, soothe inflammation which could trigger the KP.
KP’s neuroactive metabolites broadly modulate neurotransmission. The two main metabolite culprits commonly measured are kyurneic acid (KynA) and quinolinic acid (QA).
- KynA is an NMDA receptor antagonist, and decreases in blood concentrations are associated with depression.
- QA is an NMDA receptor agonist, considered neurotoxic, and higher concentrations are associated with depression.
It could be beneficial to have a high KynA to QA ratio. In a new study out of the Laureate Institute for Brain Research (LIBR), twenty healthy male- and female-identifying adults took 200 or 600 mg doses of the NSAID ibuprofen or a placebo. The hypothesis was that this anti-inflammatory would tip the synthesis pathway towards KynA over QA.
Image Credit Wikimedia Commons, via Creative Commons license CC BY 3.0, no alterations made.
This is a hot hypothesis to explore. In 2020, a multinational meta-analysis sought to settle the basic trends of the kynurenine pathways in major depressive disorder, bipolar disorder, and schizophrenia. Across these three disparate disorders, the studies found a decrease in tryptophan. Among other ratio differences, in major depressive disorder and bipolar disorder, there was a favoriting of QA synthesis over KynA, indicating the metabolism of tryptophan was redirected.
The preliminary study from LIBR found a 600 mg dose of ibuprofen increased the blood concentrations of KynA by around 25% compared to the placebo. That dose also increased the KynA/QA ratio. Both dose levels decreased tryptophan concentrations. The change in tryptophan was attributed to “being displaced from its binding site on albumin by ibuprofen” and filtered out by the renal system.
Researchers admit this study didn’t include a patient population. In non-depressed individuals from this and other studies, ibuprofen has had “marginal” or “small but significant” effects on depressive symptoms. There is also a possibility these effects are more applicable to female-gendered patients than others. Another knock to the monoamine theory being the main factor in depression, the authors conclude ibuprofen could have neuroprotective effects for depression.
Indeed, these findings add credence to the concept of depression being a neuroinflammatory disorder. The shift of pathways away from serotonin and towards the KP leads to measurable depression improvement. KP is establishing itself as a “key pathophysiological mechanism.” Though highly preliminary, the next stage of depression research will likely be with NSAIDs.
But how could little old ibuprofen be helping? NSAIDs increased KynA through unknown means, and there are multiple ways this could be occurring. NSAIDs block the activity of two enzymes responsible for kicking off inflammatory responses: cyclooxygenase 1 (COX-1) and 2 (COX-2). There would be fewer pro-inflammatory prostaglandins without the various COX. Alternatively, it could inhibit the enzyme synthesizing the neurotoxic QA metabolite and encourage the formation of the helpful KynA.
It seems like inflammation is the “in-vogue” thing killing us. Destructive to multiple organ systems, including the central nervous, inflammation seems to be an ever-present part of modern society. When the usually helpful microglia are triggered by the inflammatory element group cytokines, their release leads to:
- activation of the hypothalamic-pituitary-adrenal or stress axis
- imbalanced serotonergic and noradrenergic circuitry
- decreased production of QA
The author’s here cite research from 2015 suggesting that when taken concurrently with the frequently prescribed depression drug serotonin reuptake inhibitors (SSR1), ibuprofen can reduce measurements of clinical psychiatric need. This study had over 20,000 participants using both SSRIs and NSAIDs. It found no safety outcome issues with this combo.
But don’t be so quick to add ibuprofen to your daily pill dispenser. Conflicting safety outcomes suggest the SSRI/NSAID pairing could be dangerous. While some reviews still tout the benefits of NAIDs on some forms of major depressive disorder, “either as single treatment or as augmentative therapy” with other SSRIs, they seem to be discounting a potential side effect.
One large 2015 article in British Medicine Journal found a significantly increased risk of intracranial hemorrhaging when new antidepressant users also used NSAIDs. And let’s say you make it past the 30-day hemorrhage time point used in the study. Can augmentative therapy with both NSAIDs and SSRIs have double the benefits? Probably not.
A paper in Proceedings of the National Academy of Sciences found antagonism between these two depression treatments. Looking at the frontal cortex, NSAIDs negated SSRIs’ ability to decrease certain cytokines. Behaviorally, the number of patients responding to SSRIs decreased when taken with anti-inflammatory medications such as aspirin and ibuprofen. There’s no doubt that numerous laboratories are currently constructing new ways of understanding this contentious and potentially dangerous drug pairing.
Depression seems to be more of an umbrella term for a group of symptoms with a likely diverse array of sources. The LIBR study suggests some depressed individuals might already have the treatment for their depression symptoms bouncing around in their car’s emergency kit. How might the pharmaceutical companies take this intrusion on their billion-dollar antidepressant marketplace? Likely, not well.
The pandemic rocked the mental stability of millions of people. Worldwide, the antidepressant market size went up 28% during the first year of the pandemic. The global sales of antidepressants jumped from 11.67 billion pre-pandemic to 16.44 billion in 2022.
Further research on individuals with histories of psychiatric disorders won’t likely receive much support from this giant industry. Instead, we will rely on institutes and government funding to determine if the best treatment for some forms of depression has been in our medicine cabinets all along.
Sources: Nature, Psychopharmacology, Fortune Business Insights, Journal of Psychopharmacology, British Medicine Journal, Proceedings of the National Academy of Sciences, Brain and Behavior