Resilience and susceptibility to depression are a complex mix of many factors. Understanding why some individuals are resilient could lead to new pathways and systems to target for the treatment, or even prevention, of depression. Researchers led by Bruno Giros at McGill University have identified a specific circuit that confers resilience to social defeat-induced depression.
Dopamine neurons in the ventral tegmental area (VTA) have long been know to play a role in stress resilience. The VTA is a crucial part of the limbic system, colloquially known as the reward pathway, a circuit that is heavily implicated in mood disorders and addiction. Previous work has shown that the dopamine (DA) neurons in the VTA are more excitable in the brains of mice that are susceptible to depression.The research presented in this recent
Nature Neuroscience paper details the noradrenergic control of the DA neurons in the VTA as a crucial factor in determining resilience to social defeat-induced depression.
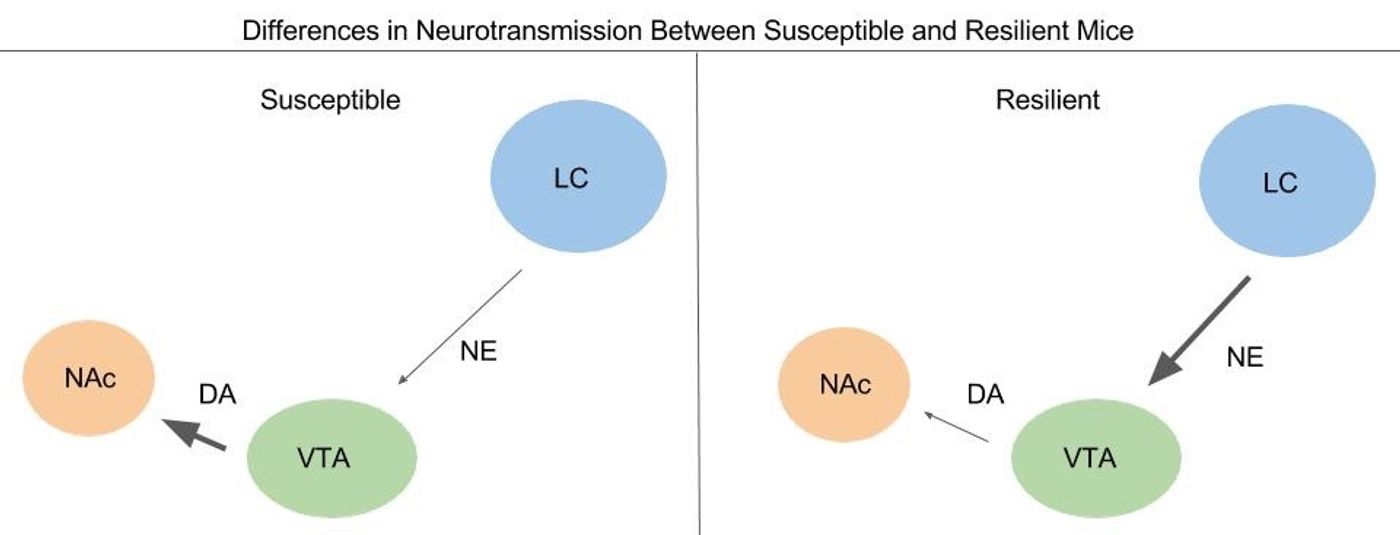
After 10 days of chronic social defeat stress, mice were determined to be either resilient or susceptible to social defeat-induced depression by measure of social aversion. The mice categorized as susceptible had less noradrenergic tissue in the VTA compared to the resilient mice, or wild type mice not exposed to social defeat stress. (Note: noradrenaline = norepinephrine, abbreviation is NE). The NE neurons originate in a region called the locus coeruleus (LC) and project to the VTA. The susceptible mice had less NE being released in the VTA and had more DA neuron excitability while the resilient mice had more NE and less DA activity. Specifically, the DA neurons in the VTA that exhibited more bursting activity in the susceptible mice were projecting to the nucleus accumbens (NAc), not the medial prefrontal cortex (mPFC). Both the NAc and mPFC are part of the limbic system. From this, the researchers concluded that resilience to depression is determined by the level of NE-mediated inhibition of DA activity in the VTA.
To confirm this further, they tested this idea pharmacologically using an α2 noradrenergic receptor antagonist which has been shown to increase activity in the LC and increase NE release. Interestingly, chronic treatment with this antagonist reversed the susceptible phenotype in the mice, but acute treatment did not. The researchers also tried a selective NE reuptake inhibitor, similar in action to an SSRI, and again observed that chronic treatment made susceptible mice resilient, but acute treatment had no effect. A well-known issue with SSRIs is that they take weeks to exert their antidepressant effects, and it appears that the effects of increasing NE release in the VTA undergo a similar latency.
The main conclusion of the paper is that NE neurotransmission from the LC to the VTA is necessary and sufficient to confer resilience to stress-induced depression. The sheer amount of experiments and techniques used in this paper, many of which have not been summarized here, makes this finding very convincing. Hopefully, this paper will lead to increased research on and production of NE-focused antidepressant treatments, and also investigations into why the NE reuptake inhibitors and SSRIs have such a long lag time between drug treatment initiation and antidepressant effects.