Autism is a mystery because it has been very difficult to narrow down what causes this complex and varied pathology. Mutations in various genes have been implicated as risk factors and one such gene is
Shank3. The SHANK3 protein is a scaffold protein that is expressed ubiquitously, but is heavily enriched in the postsynaptic densities of excitatory synapses. Deletion of
Shank3 is thought to be one of the principal causes of Phelan-McDermid syndrome. Phelan-McDermid syndrome (PMS) is a genetic disorder caused by the deletion of a portion of chromosome 22, where the
Shank3 locus is. The main symptoms of PMS are thought to be a result of Shank3 deletion and include developmental delay, intellectual disability, and absent or delayed speech. PMS patients can also exhibit autistic-like behavior such as limited eye contact, adding to the support for SHANK3’s involvement in autism spectrum disorders (ASD).
It is possible that loss of SHANK3 function causes these impairments simply through impaired synaptic function, but some of the cellular changes in
Shank3-mutant neurons challenge this explanation. A recent paper by first authors Fei Yi and Tamas Danko from Stanford published in
Science details the cellular mechanism behind the mutant
Shank3 phenotype. Using neurons derived from human embryonic stem cells with a
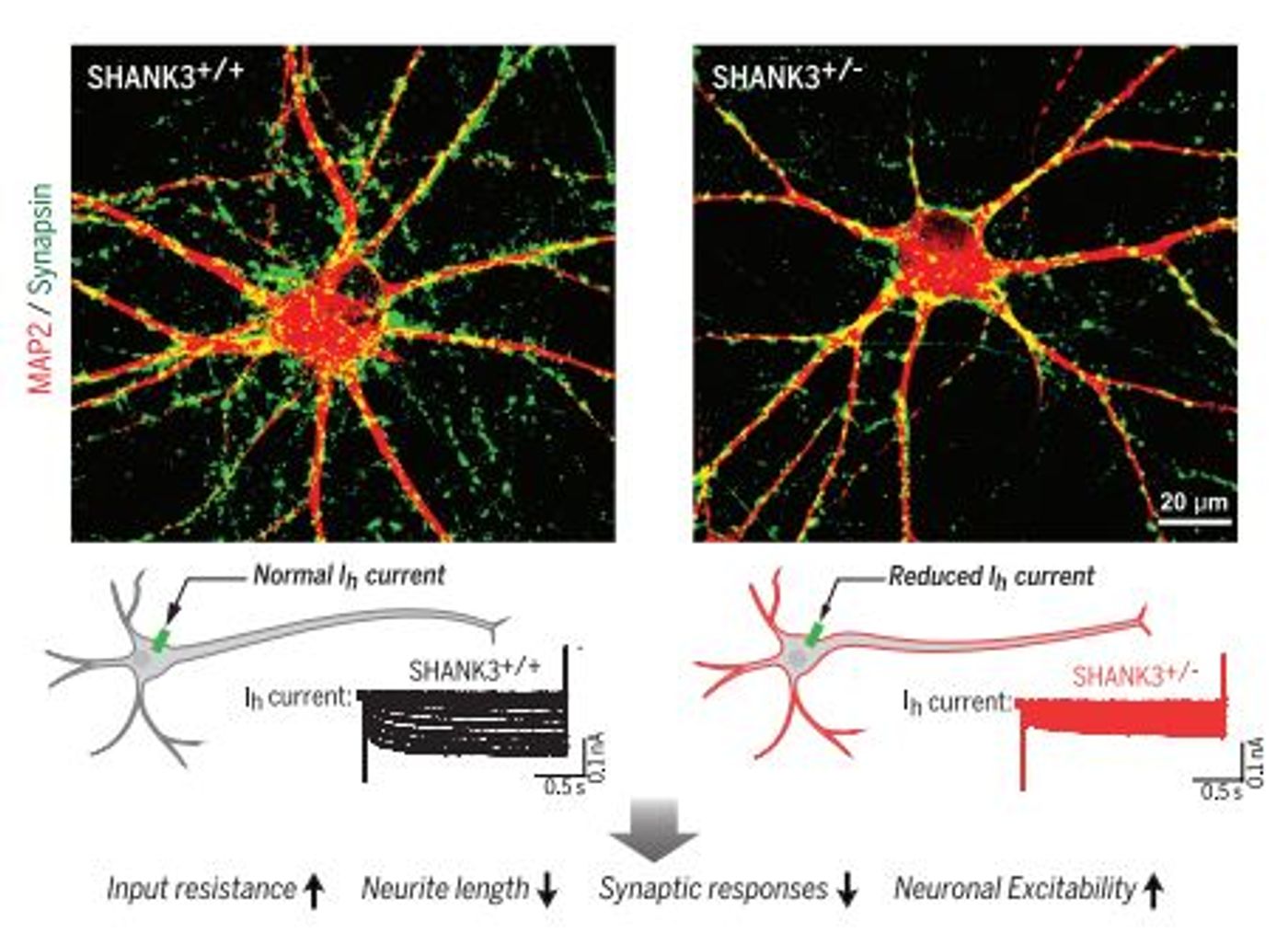
Shank3 conditional knockout, the researchers saw that insufficient SHANK3 led to a wide variety of cellular changes, including synaptic deficits and altered electrical properties. The question then becomes how does loss of a postsynaptic scaffolding protein lead to changes in the intrinsic electrical properties of the neuron?
The main electrical property change that they saw was a huge increase in input resistance. In this context, the increased input resistance makes the neurons more excitable in response to the same amount of current. From this observation, they hypothesized that altered channel conductance is the primary impairment caused by insufficient SHANK3. What they found is that insufficient SHANK3 protein causes a huge impairment in a particular cation current called I
H. The I
H current goes through particular channels called HCN channels and it is a particularly interesting current because it is activated by hyperpolarization. The resting potential of a neuronal membrane is ~ -70mV and the neuron fires when the membrane is depolarized to ~ 35mV. Hyperpolarization is when the membrane potential is even more negative. At this potential, the I
H current lets cations (positively charged ions) into the neuron to depolarize the membrane back to its resting potential. Because of this functionality, HCN channels and I
H currents are known as “pacemakers”; they help to establish rhythmic or oscillatory firing in neurons. HCN channels may also play a role in synaptic plasticity. Pharmacological blockade of I
H currents mimics the cellular phenotype of insufficient SHANK3, confirming that the changes in neuronal electrical properties is the primary cellular phenotype caused by SHANK3 insufficiency. The other cellular changes (impaired synaptic transmission, deficits in dendritic branching, and increased excitability) are caused by the impaired I
H current. These changes were rescued by re-expression of functional SHANK3.
It is important to figure out the cellular changes caused by SHANK3 insufficiency because it is a gene that has been heavily linked to autism. The research presented in this
Science paper, apart from being super interesting, could eventually lead to a pharmacological target to relieve some of the symptoms of PMS and ASD.
Sources:
Science,
PMS, and
HCN review