Immunotherapy Diversification: From CAR-T cells to CAR-NK cells
Immunotherapy is an advancing field within the scope of therapeutic oncologic approaches. It is of great interest because it aims to utilize the human body’s own immune system to thwart and eradicate cancerous cells. The discovery of chimeric antigen receptor (CAR) models to augment anti-tumor actions by our natural immune system was actually made in 1891 by a New York surgeon named William Coley. His use of an injected beta-hemolytic Streptococcus species into a patient with osteosarcoma demonstrated that enhancing the immune system can result in tumor shrinkage when the conditions and factors involved are right. While at the time the mechanism was unknown, what became known as “Coley’s toxins” were really the beginnings of immunotherapy. Coley had noticed that patients with cancer who got infections after surgery did better than patients without infection. His hypothesis was that the infection promoted or enhanced the immune system’s natural function and resulted in better cancer outcomes. There were others at the time that utilized his method successfully, it has been reported that even the Mayo brothers communicated with Coley about their results, but it did not always produce the desired results; Coley documented that each injection was painstakingly made for each individual patient and others, who tried to replicate is work, were unsuccessful perhaps because less care was taken for each individual injection, as has been suggested in the literature. Now, 100+ years later, scientists are reviving his theories after they were discarded because replication attempts by others failed.
CAR-T cell therapy has been on the forefront of cancer immunotherapy research and some therapies have even come far enough along to be approved by the United States Food and Drug Administration (FDA) for clinical patient use. Still other areas of CAR-T cell therapy are being investigated for other types of cancers and are in the midst of clinical trials.
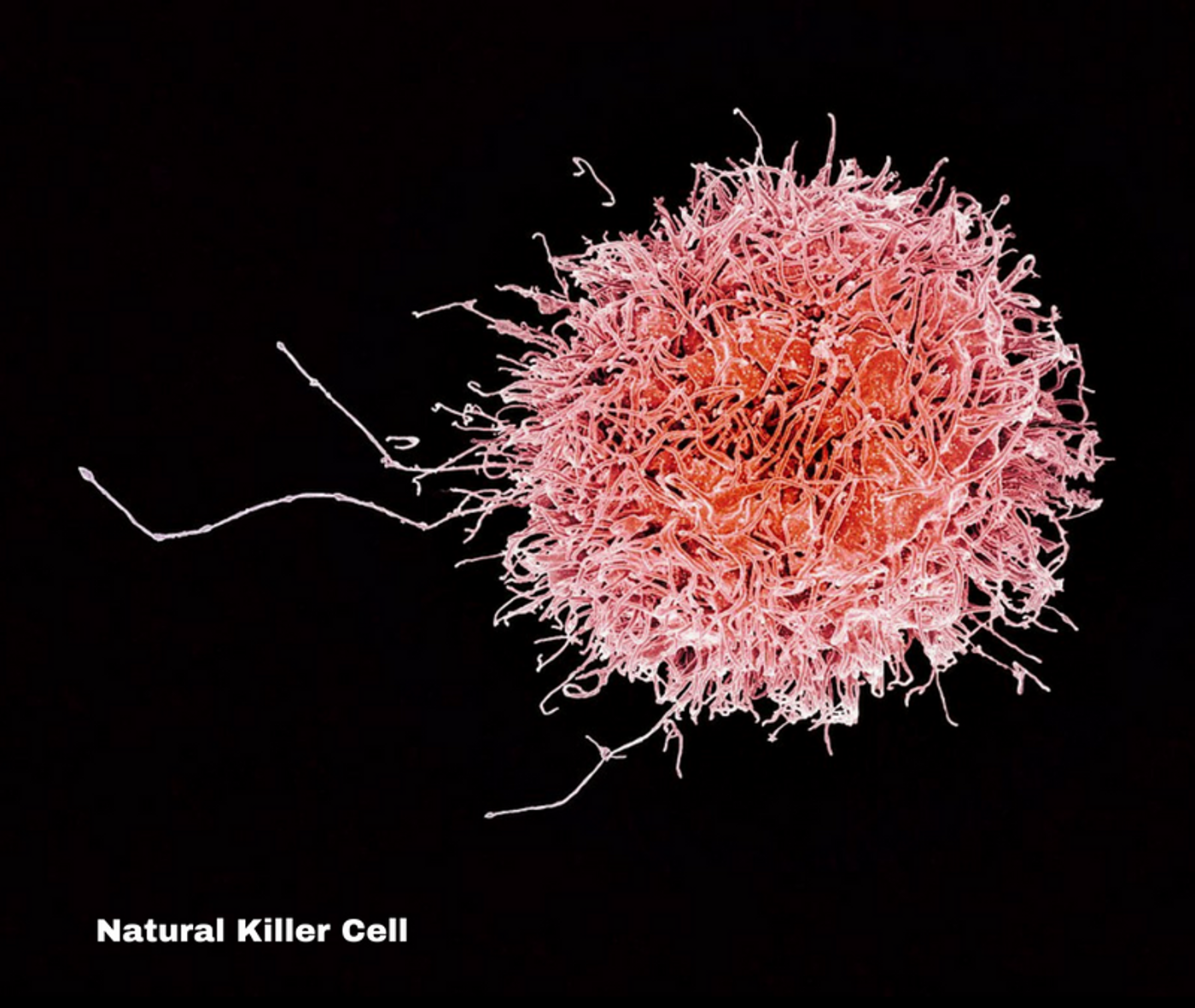
A new delivery design that utilizes the CAR model has just jumped onto the map and is reportedly being researched at MD Anderson, the University of San Diego School of Medicine, and the University of Minnesota. This new immunotherapy utilizes natural killer cells or NK cells along with the CAR motif to destroy tumor cells. NK cells are part of the body’s innate immune system. They are lymphocytes and come from a progenitor cell shared in common with T and B cells. NK cells respond quickly to diverse pathogens encountered within the body. NK cells are the body’s normal cancer fighting reserve which is how they got their namesake. This ability to kill cancerous cells arises when a cell is determined to be in need of destruction either because of errors in cell division or DNA replication that results in mutagenic expression. Before the mutagenic expression impacts other cells or replicates itself, NK cells recognize the cell and destroy it. They also recruit other cells, macrophages and dendritic cells, to enhance an immune response and phagocytize cellular material that remains where necessary.
Researchers are using these NK cells and CARs to create CAR-NK cells which is exciting for a few reasons. The first is that because NK cells are already patrolling for particular cell expression that looks mutagenic, they don’t need the same type of antigen presenting priming like CAR-T cells do for each individual cancerous antigen. This means that these NK cells can be used for multiple different cancers and multiple different patients with potentially similar results. Secondly, the toxicity of this type of treatment has less potential for long term damage or late stage effects because they mobilize the innate immune system already in place in the body. The research organizations working on this new direction in immunotherapy have different methods of generating NK cells.
The University of Minnesota and University of San Diego are collaborating on the project and utilize human induced pluripotent stem cells (iPSCs) to generate NK cells. They can take skin cells or blood cells and provide signals to revert them back to their pluripotent stem cell state, meaning they can become whatever cell is needed simply through use of the appropriate signaling molecules, and then induce them to become NK cells, all in a laboratory setting. The idea is that these can be “ready-made” therapeutic treatments which avoid the constraints that come with patient specific therapeutic approaches. The collaborative group is looking to take their delivery model to clinical trials in the coming months and reported very few adverse side effects in the mouse models used so far.
MD Anderson researchers are studying CAR-NK cell therapies but use cord blood and separate out the NK cells. They insert a CAR that recognizes CD19 and is currently in clinical trials that started last year (NCT03056339). Patients that participate have to have relapsed disease and are focusing on B-Lymphoid malignancies, Non-Hodgkin Lymphoma, and both Acute and Chronic Lymphocytic Leukemia.
Sources: Cell Stem Cell, Experimental Biology and Medicine, Cancer Research Institute, ClinicalTrials.gov, Healthline,