Overall brain cancer is rare, but it’s actually the most common form of solid tumors in children under 15 years old. Chemotherapy is not always effective, and in some cases it leaves children with lifelong side effects even if they do survive the cancer. On a mission to improve pediatric brain cancer diagnoses to make treatments safer and more specialized, scientists from the Perelman School of Medicine at the University of Pennsylvania, The Children's Hospital of Philadelphia (CHOP), and the Dana Farber Cancer Institute investigated the genetic drivers behind the development of these rare cancers.
Pediatric low-grade gliomas are the most common type of pediatric brain cancers, and most of these cancers result from a mutation in the mitogen-activated protein kinase (MAPK) pathway. This series of genes provide functions for the cell like proliferation, differentiation, motility, stress response, apoptosis, and survival (
Cell Signaling Technology). There are drugs being developed to specifically target MAPK-related brain tumors, but the collaborative team from this study decided to look at the rare tumors not related to MAPK mutations. The study was published in the journal
Nature Genetics.
The study, led by Phillip Storm, MD, and Adam C. Resnick, PhD, centered on the genetic information from 249 non-MAPK pediatric brain tumors. The most common gene mutation from this sample was a mutation in the myeloblastosis (MYB) gene, with 19 out of 19 angiocentric gliomas having the same MYB mutation. Angiocentric glioma is a slow-growing tumor recognized a distinct type of cancer by the World Health Organization in 2007 (
Journal of Neurosurgery: Pediatrics).
The mutation was a chromosome deletion fusing MYB to another, usually separate gene, called quaking homolog, KH domain RNA binding (QKI). The team of researchers saw this fusion driving tumor formation in three different ways.
- MYB expression in the brain
- QKI unable to suppress tumors despite its programming to do so
- MYB-QKI fusion drives its own expression
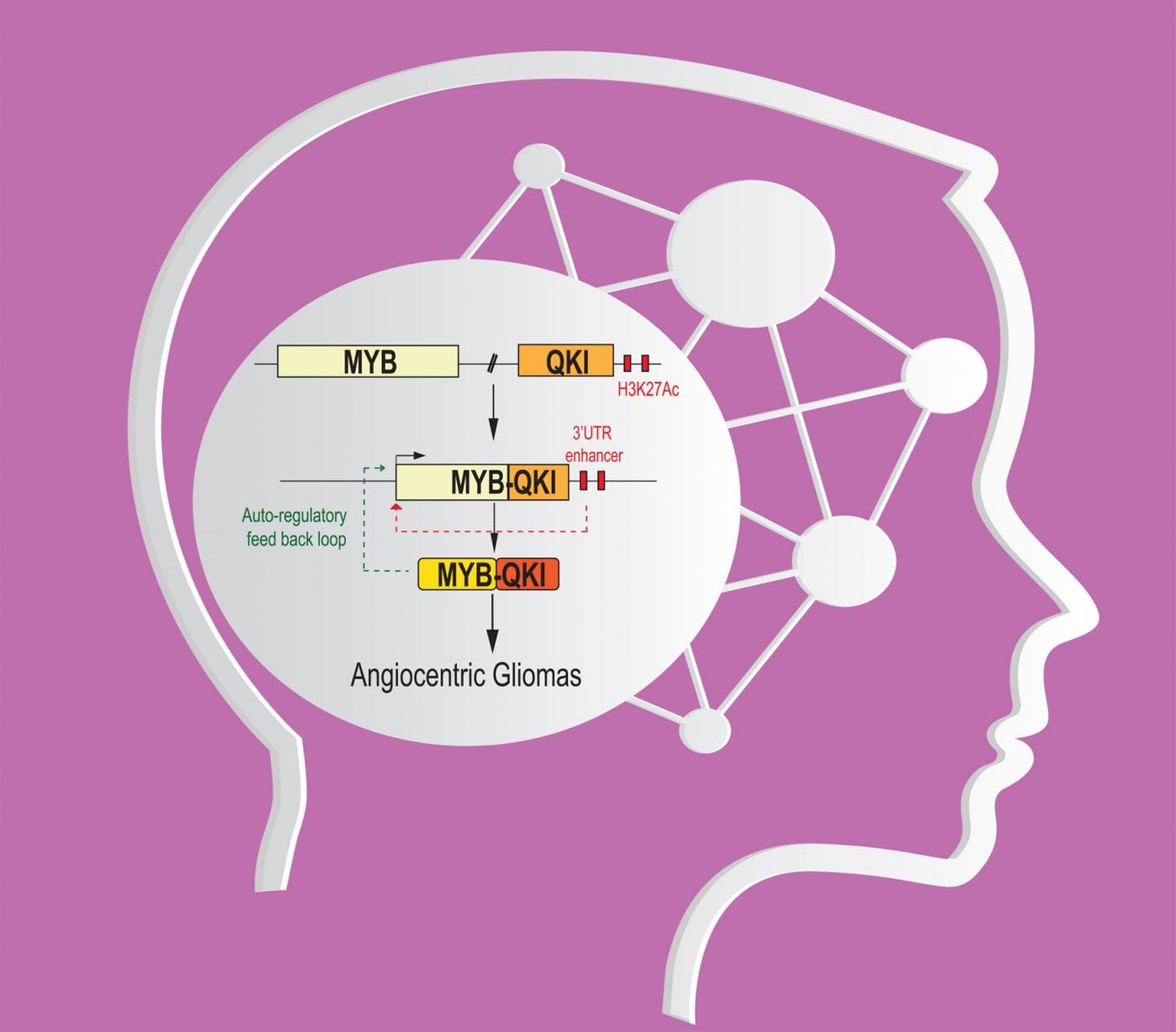
This dangerous trio of unfortunately random combinations creates a dangerous tumor. In addition, the problem is exacerbated when the cells attempt to increase QKI production to prevent abnormally growing cells. With MYB fused to QKI, not only can QKI not perform its suppressive duties, increasing production of the fused genes only makes the tumor larger. The MYB-QKI gene fusion also tested positive in mice for tumor creation.
Although uncovering the mechanisms behind this rare cancer revealed a dangerous mutation wreaking havoc on the cells of the brain, scientists can now help doctors determine between MAPK-related and MYB-QKI-related brain cancer. There is no treatment specifically for MYB-QKI tumors yet, but recognizing this distinction can prevent unnecessary MAPK-targeted chemotherapy.
"Gene fusions are common in cancers, and we think it's likely that some will be found to work via combinatorial mechanisms like those we've uncovered in this study," Resnick said.
In the future, Resnick, Storm, and the rest of their team plan to investigate the possibility of blocking or mending the epigenetic changes that lead to gene fusion-related pediatric brain cancers.
Source:
University of Pennsylvania School of Medicine









