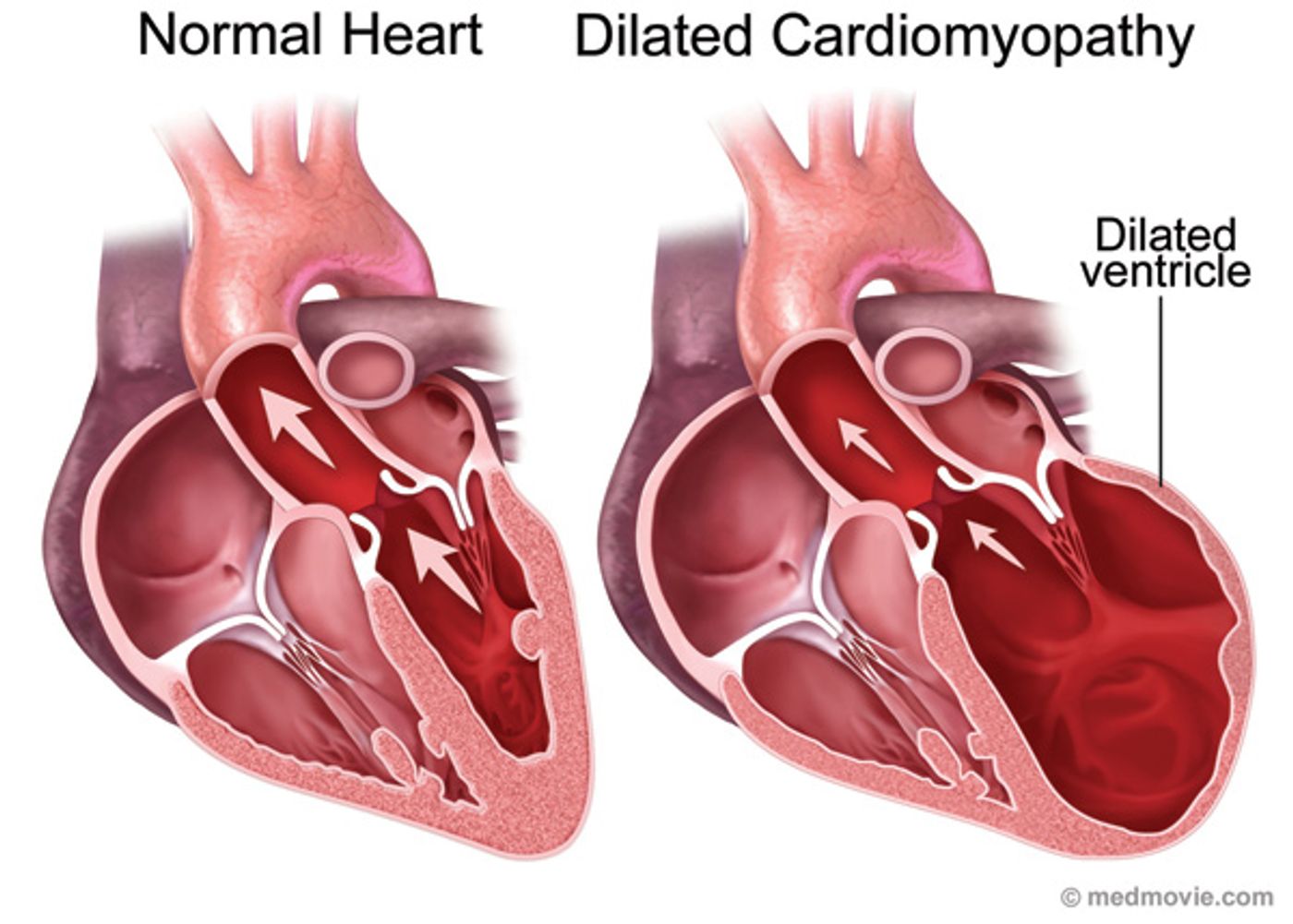
Dilated cardiomyopathy (DCM) and hypertrophic cardiomyopathy (DCM) share some qualities in their tissue makeup and in their pathophysiology, but HCM usually does not progress to heart failure, while DCM does. In a new study of transcriptional and molecular changes during DCM development, scientists from Harvard Medical School and the Emory University School of Medicine uncover genetic secrets behind the progression of DCM and its differences from HCM.
DCM is characterized by a rapid thinning of heart muscle cells, called cardiomyocytes, often inherited from parents but also appearing as a secondary complication from an infection or from alcohol abuse. DCM usually transitions from a pre-stage to diseased stage and finally heart failure, and about 1 in 250 individuals are affected. Functional and structural changes that occur as DCM progresses all stem from an initial inhibition of regular myocardial calcium homeostasis.
Only occurring as a genetic disorder, HCM similarly stems from irregular myocardial calcium homeostasis. However, in the present study, scientists show that DCM has a specific “genetic signature” that clearly distinguishes how the two heart diseases progress so differently.
Using RNA sequencing to complete longitudinal transcriptome analysis, researches compared the gene expression profiles of cardiomyocytes and non-myocytes during different stages of disease. Additionally, they compared these genetic profiles to those of similar cells in HCM cases.
They used a genetic mouse model of inherited DCM that carried a “phospholamban missense mutation” to mimic the DCM state of altered myocardial calcium homeostasis, causing DCM to develop in the subjects.
Signs of early DCM progression were clear as the researchers examined the results. Increased expression of fibrotic genes, of inflammatory genes, and cardiomyocyte metabolic changes all appeared to have an impact on DCM developing initially as well as eventually progressing into heart failure. Metabolic changes included reduced expression of genes for aerobic respiration and increased expression of genes for glucose utilization.
“The results of this study provide new insight into the gene programs that drive cardiac remodeling,” the scientists said. The study was recently published in
JCI Insight.
As they followed the stages of disease throughout the study, the scientists noticed that while hearts in the preliminary stage appeared normal, more in-depth analyses showed an enhanced proliferation of nonmyocytes, 60 percent more (than normal controls) to be exact. Additionally, these pre-DCM hearts showed enhanced activity of both proinflammatory signaling and profibrotic cytokines specific to cardiomyocytes. As DCM progressed further, almost 20 percent of the left ventricle would become fibrotic.
This study significantly showed both the difference between DCM and HCM progression as well as uncovered the genetic signature that was responsible for the myriad of changes that occur as this disease develops. In fact, the same markers that characterized early progression of DCM into heart failure also helped to distinguish diagnoses of DCM versus HCM. Scientists hope to continue studying this genetic signature in hopes that further research will reveal targets for treatment.
Source:
JCI Insight