Risk factors leading to cardiovascular disease are often thought to be obesity, a sedentary lifestyle, and environmental factors like smoking habits and diet. However, a new study from the Salk Institute goes much deeper to the nuclear level, finding molecular evidence on the nucleus’ involvement with the changes the lead to diseases of the heart as well as leukemia and aging disorders.
“Our research shows that, far from being a passive enclosure as many biologists have thought, the nuclear membrane is an active regulatory structure,” said Salk professor Martin Hetzer. “Not only does it interact with portions of the genome to drive gene expression, but it can also contribute to disease processes when components are faulty.”
Traditionally, scientists believed the role of the nuclear membrane to be chiefly to keep the nucleic contents, the DNA, physically separated from the rest of the cell with a protective enveloping membrane. Known complexes of thirty or more proteins, called nucleoporins, poke holes in the membrane to become gatekeepers for molecules wanting to exit or enter the cell. However, the new Salk study shows that the nuclear membrane has a dual role: acting on the genetic material contents to influence gene expression.
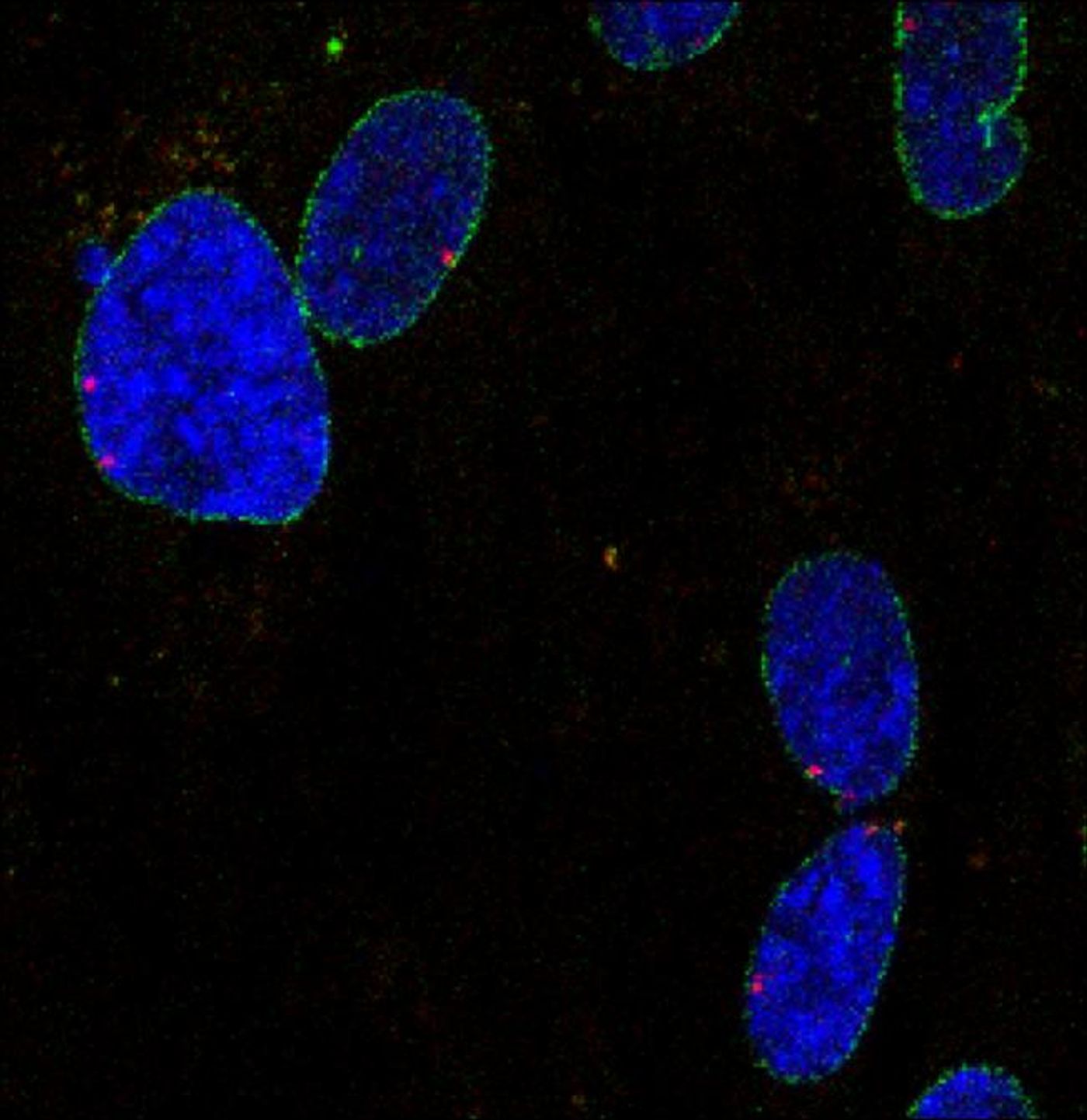
Salk scientists began their study with a human bone cancer cell line to study in detail the regions of the DNA that potentially experienced interaction with the nucleoporins. A novel molecular biology technique called DamID was used to successfully identify two specific nucleoporins – Nup153 and Nup93 – that directly interacted with the genome. Going further, the scientists used advanced sequencing techniques to see which specific genes the two nucleoporins were interacting with and how the interaction changed gene expression.
“Nuclear pore proteins are critical for the function of those genomic sites,” explained first author Arkaitz Ibarra.
Indeed, they found that Nup153 and Nup93 form complexes spanning across the entire surface of the membrane, actively regulating the expression of key genes, thought to ultimately influence the development of cardiovascular disease, leukemia, and aging disorders. This finding was, according to Ibarra, “very unexpected.”
Specifically, Nup153 and Nup93 were found to interact with and regulate expression of specific parts of the genome known to aid in determining cell identity, called super-enhancers. In studies of silenced Nup153 or Nup93, abnormal gene expression from the associated region followed either way. Salk Institute scientists confirmed their findings with a lung cancer cell line that showed nucleoporins interacting with several super-enhancer regions to influence gene expression. Moreover, experiments that altered the NPC proteins made related gene expression faulty even though the proteins still performed their primary role as gatekeepers in the cell membrane.
Ibarra and the team from Salk believe that their results could provide monumental insight into why diseases like heart disease, leukemia, and aging disorders show connections to dysfunctional nuclear membrane components. Knowing now that specific nucleoporins associate with DNA and influence gene expression, scientists could look further into their dysfunction as a potential cause of disease.
“People have thought that the nuclear membrane is just a protective barrier, which is maybe the reason why it evolved in the first place,” Ibarra said. “But there are many more regulatory levels that we don’t understand. And it’s such an important area because so far, every membrane protein that has been studied and found to be mutated or mis-localized, seems to cause a human disease.”
The present study was published in the journal
Genes & Development.
Source:
Salk Institute