Getting Rid of Damaged Mitochondria Relieves Autoimmune Disease in Mice
Inflammation is a vital process in the body. It helps protect us from invading pathogens and helps us heal after wounds occur. But when inflammation gets out of control, it can cause significant harm and lead to many types of chronic disease. One way the immune system can keep the process regulated is through protein complexes called inflammasomes. The one we might know the most about is the NLRP3 inflammasome, which is active when mitochondria, the cell’s powerhouses, are stressed or have been damaged. An overactive NLRP3 inflammasome has been linked to a variety of chronic inflammatory disorders. New work reported in Cell Metabolism has shown that in mice, cells can be induced to eliminate damaged mitochondria before the NLRP3 inflammasome is activated.
The leader of this work, senior author Michael Karin, Ph.D., a Distinguished Professor of Pharmacology and Pathology at UC San Diego School of Medicine, reported in 2018 that damaged mitochondria can activate the NLRP3 inflammasome. In a process called mitophagy, cellular recycling machinery removes damaged mitochondria. The NLRP3 inflammasome, the researchers found, is deactivated during that process.
"After that, we wondered if we could reduce harmful excess inflammation by intentionally inducing mitophagy, which would eliminate damaged mitochondria and should in turn pre-emptively inhibit NLRP3 inflammasome activation," explained Karin. "But at the time we didn't have a good way to induce mitophagy."
A postdoctoral researcher in the Karin lab, Elsa Sanchez-Lopez, Ph.D., was investigating how cells control the uptake of a nutrient called choline. She found that an inhibitor of an enzyme choline kinase or ChoK is able to initiate mitophagy. With ChoK inhibited, choline is not added to mitochondrial membranes, and cells can sense that mitochondria are damaged. The mitochondria are then disposed of, through mitophagy.
"Most importantly, by getting rid of damaged mitochondria with ChoK inhibitors, we were finally able to inhibit NLRP3 inflammasome activation," Karin added.
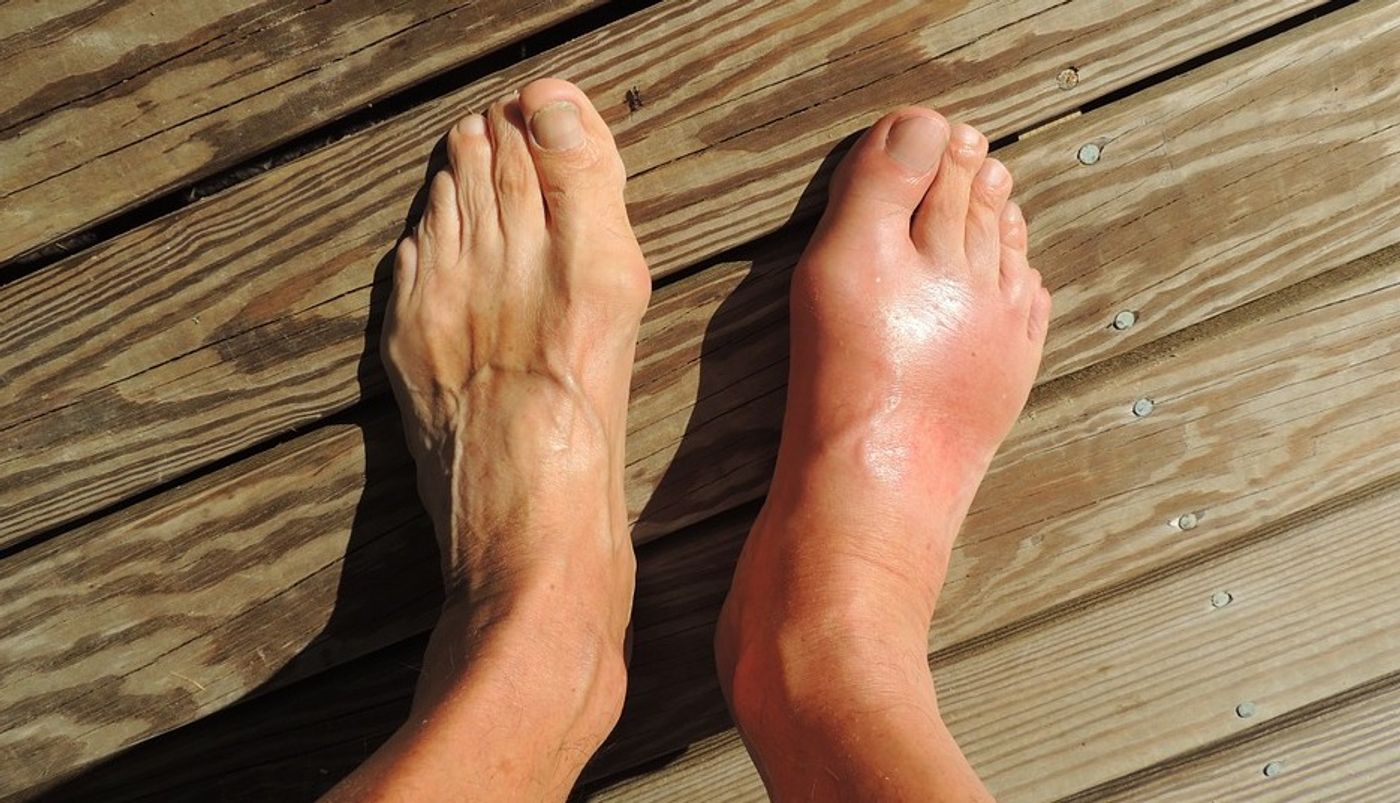
The scientists used a mouse model to test the effects of ChoK inhibitors. Treatment with the inhibitor stopped acute inflammation when the mice were exposed to uric acid, which mimics gout flares. The ChoK inhibitor also reversed symptoms of chronic inflammation seen in a model of Muckle-Well Syndrome, a genetic disorder caused by NLRP3 gene mutations. Enlarged spleens are seen with increased inflammation, and in Muckle-Well Syndrome mice, spleens are about twice the normal size. After treatment with ChoK inhibitor, the spleens returned to normal size.
While the NLRP3 inflammasome exerts its effects through two cytokines (IL-1β and IL-18) that promote inflammation, existing drugs only block one of those two cytokines. ChoK inhibitors, however, block both cytokines.
"There are several diseases, including lupus and osteoarthritis, whose treatment will likely require dual inhibition of both IL-1β and IL-18," Karin explained.
Learn more about the inflammasome from the video.
Sources: AAAS/Eurekalert! Via UCSD, Cell Metabolism