Dysfunctional Mitochondria Cause Telomere Damage
Reactive oxygen species (ROS) are a natural byproduct of many biological processes and are considered normal signaling molecules. The energy-generating powerhouse of the cell, the mitochondria, is also a source of ROS. But ROS can cause damage to DNA and cell membranes and can promote inflammation. New work has now shown that damaged mitochondria can produce a wave a ROS that has a cascade of negative impacts on the cell. Damaged mitochondria have been connected to a variety of diseases. The findings, which show how sick mitochondria cause more cellular damage, have been reported in the Proceedings of the National Academy of Sciences (PNAS).
"I like to call it 'the Chernobyl effect' -- you've turned the reactor on and now you can't turn it off," said senior author Bennett Van Houten, Ph.D., professor of pharmacology and chemical biology at the University of Pittsburgh School of Medicine and UPMC Hillman Cancer Center. "You have this clean-burning machine that's now polluting like mad, and that pollution feeds back and hurts electron transport function. It's a vicious cycle."
A technique created by Marcel Bruchez, Ph.D., of Carnegie Mellon University was used in this study. It enabled Van Houten's team to induce the production of ROS in response to light.
"That's the Chernobyl incident," Van Houten said. "Once you turn the light off, there's no more singlet oxygen anymore, but you've disrupted the electron transport chain, so after 48 hours, the mitochondria are still leaking out reactive oxygen -- but the cells aren't dying, they're just sitting there erupting."
Related: Getting Rid of Damaged Mitochondria Relieves Autoimmune Disease in Mice
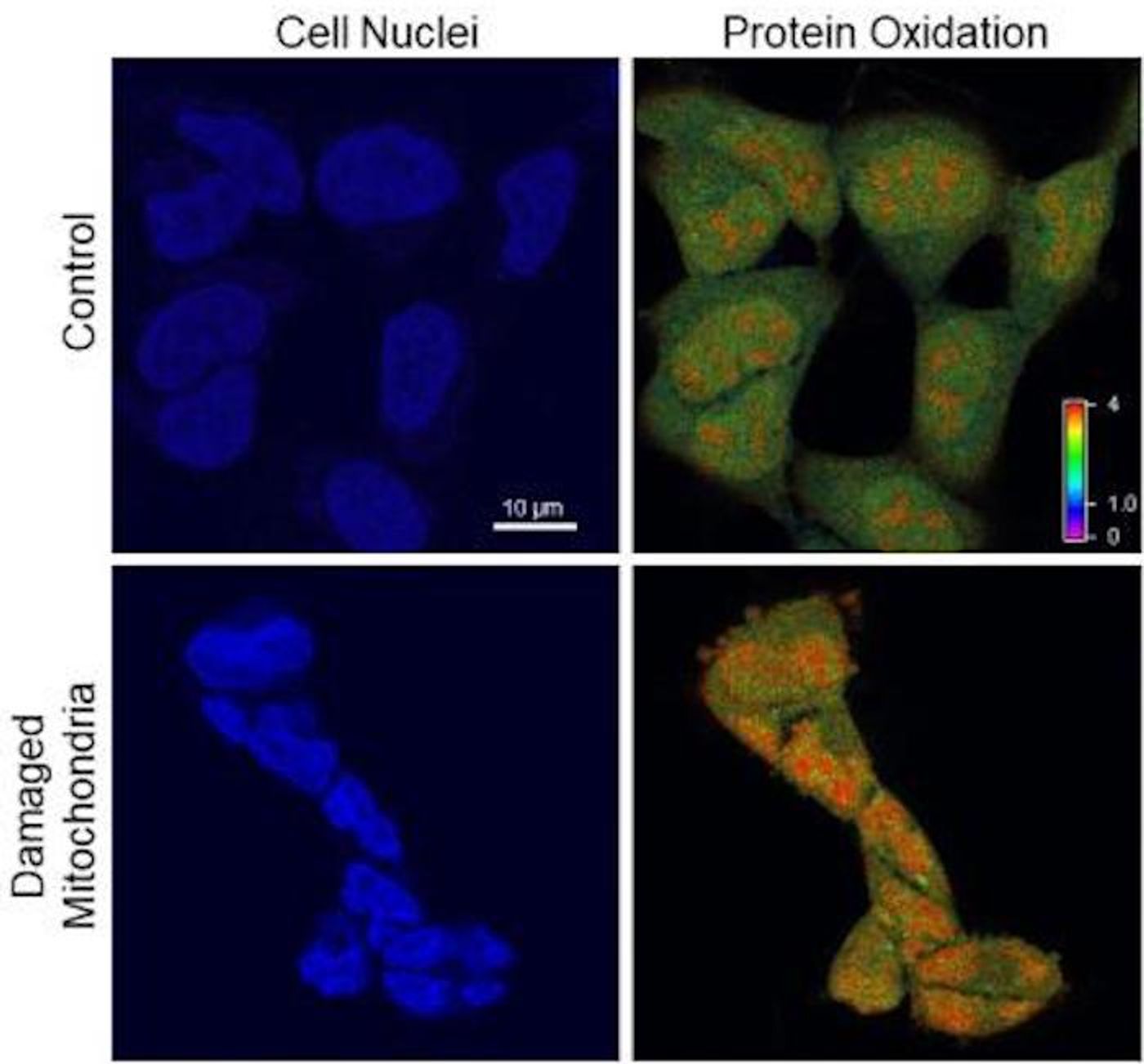
The cell stops dividing as free radicals bombard the nucleus, which starts shrinking. The DNA still seemed to look normal, until the researchers focused on the caps that protect the ends of chromosomes, called telomeres. This damage was limited to a specific area and was probably hard to find when looking at the whole genome, as the research team had been doing.
"If you imagine the chromosome as a car, the telomere would be the width of its license plate," noted study co-author Patricia Opresko, Ph.D., professor of environmental and occupational health at the Pitt Graduate School of Public Health and UPMC Hillman.
The team illuminated the ends of chromosomes with fluorescent tags, illustrating the telomere damage. The researchers then repeated the experiment but didn’t activate the mitochondria; the reaction then stopped, and free radicals weren’t building up and causing damage to the telomeres.
"Basically, we shut the machine off before it had a chance to do any damage," Van Houten explained.
This work may improve photodynamic cancer therapeutics, which use ROS to disrupt tumors. A protein called ATM also damages DNA, and when ATM was inhibited, the ROS impact was magnified; it killed the cells. It may be possible to kill cancer cells with light, suggested Van Houten, if photodynamic therapy is combined with ATM inhibition.
Sources: AAAS/Eurekalert! via University of Pittsburgh, PNAS