Understanding an Unusual Receptor That's Linked to Depression
Clinical depression is estimated to impact over 264 million people worldwide in any given year. Depression is difficult to treat, in part because we don't know the biological mechanisms underlying the issue. Researchers have now used cryo-electron microscopy to elucidate the structure of a mysterious brain receptor that has been linked to depression and anxiety, called GPR158, as well as a complex that regulates it. This work could help scientists develop more effective treatments for depression, anxiety, and mood disorders. The findings have been reported in Science.
Previous work by lead author of this study, Kirill Martemyanov, PhD and colleagues indicated that in people diagnosed with major depressive disorder, levels of the GPR158 receptor in the prefrontal cortex are abnormally high. In mice exposed to chronic stress, GPR158 levels in the prefrontal cortex also increased, and the animals exhibited depression-like behavior. When GPR158 was eliminated from mice under chronic stress, they became resistant to depression.
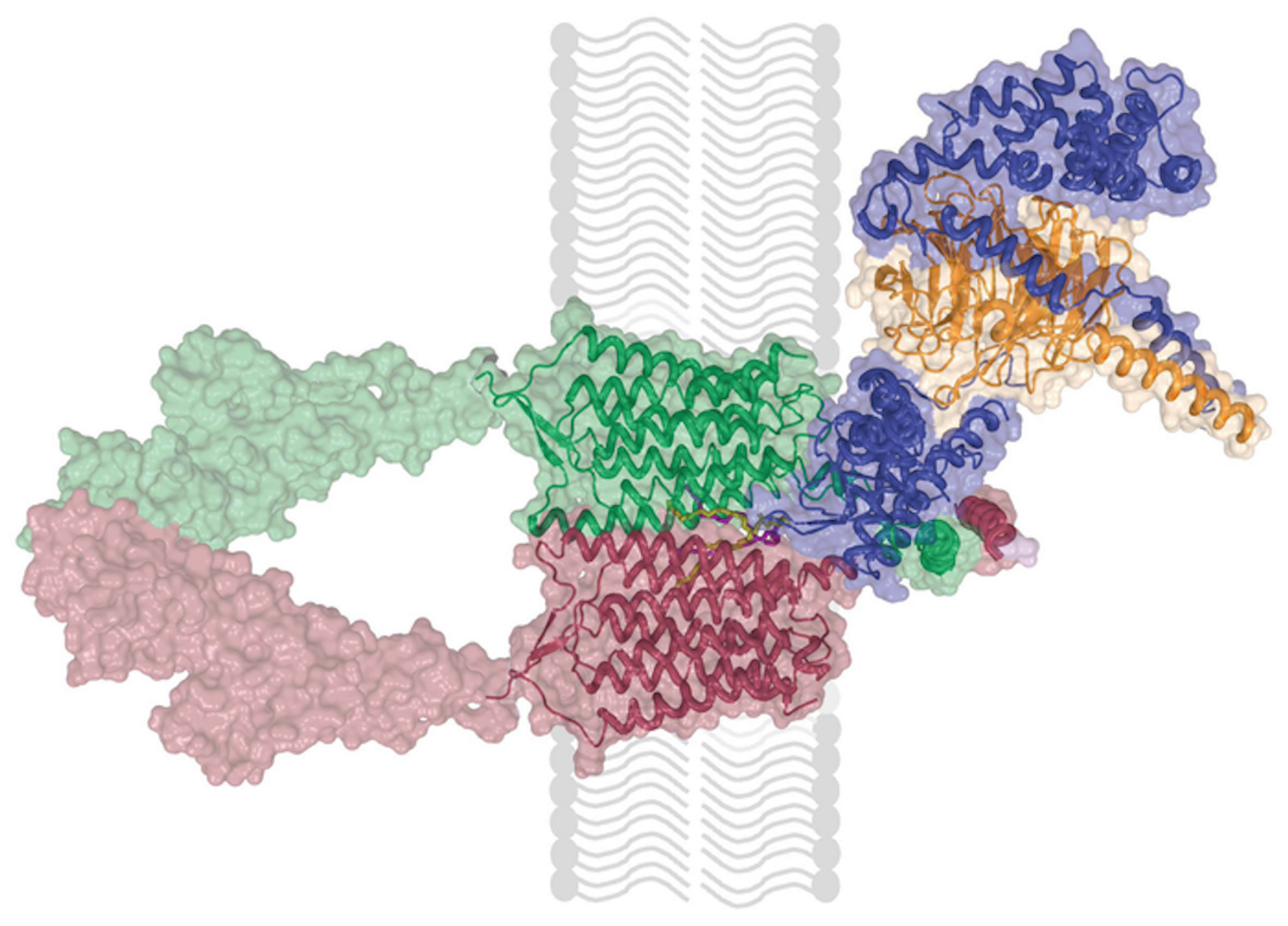
GPR158 has been known as an orphan receptor because its binding partner, which activates it, is not known. It is associated with a regulator of G protein signaling (RGS) complex and can inhibit signaling in cells. But the functional link between that RGS and GPR158 has also been a mystery.
This work has now suggested that GPR158 binds to an RGS complex to transmit a signal. It also showed that the receptor is structured as two GPR158 molecules that are linked by phospholipids. In additon, the researchers identified a kind of trap called a cache domain that may hold molecules that activate GPR158.
Now that researchers know more about the unique biology of this receptor, they can see if parts of it are potential therapeutic targets. It may be possible to break up the arrangement of GPR158, disrupt its link to the RGS complex, or aim for the cache domain, for some examples, to treat depression, although a lot more work will be needed before that happens.
Whatever strategy might be devised, the structural data on GPR158 could accelerate drug discovery and lead to new depression treatments, suggested Martemyanov, a Professor and Chair of the Department of Neuroscience at the Scripps Research Institute.
Cryo-EM was crucial for this work. “The microscope uses a beam of electrons instead of light to image protein assemblies. The shorter wavelength of electrons compared to light allowed us to visualize our sample at near-atomic resolution,” explained structural biologist Professor Tina Izard, PhD.
Sources: Scripps Research Institute, Science