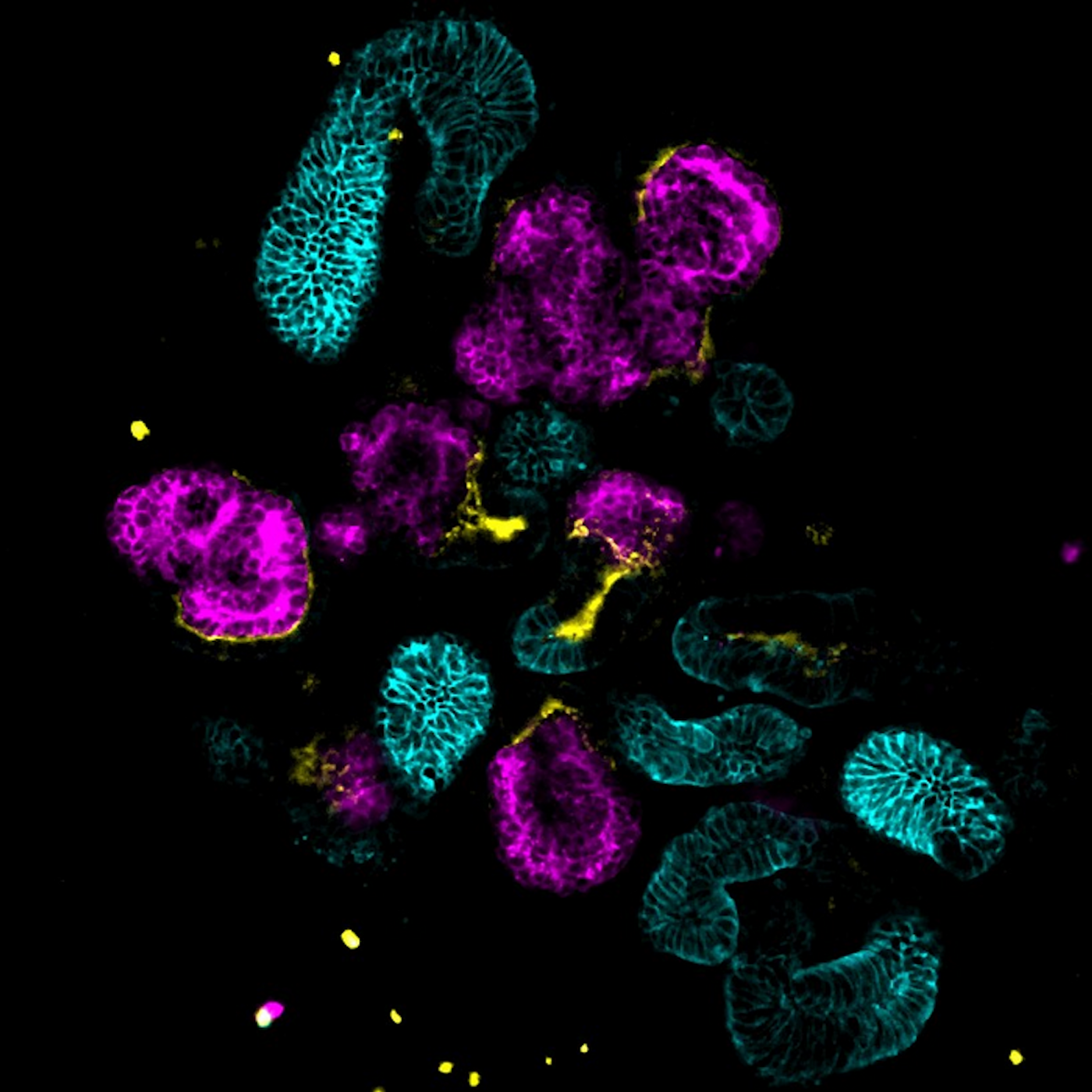
Mini-Kidneys Reveal New Insights, Treatments for Multiple Disorders
Researchers have been able to use animal models to study human disease, but there are clear limitations to that approach. Cell lines that are derived from human samples can be used as well, but many of those cells are grown in dishes - two-dimensional environments that are not like the human body. So researchers used genetic manipulation to create orgnaoids, which are miniature, simplified models of human organs. These three-dimensional structures are typically made of different cell types that express certain genes, and mimic the function of a natural organ. Scientists have now used kidney organoids to make major advances in our understanding of two different kidney disorders.
One study involved tuberous sclerosis complex (TSC), a rare disease that causes benign tumors to grow in the brain, heart, lungs, kidneys, or skin of children and adults. The disorder can vary widely from one patient to another, and may be mild or life-threatening. About 70 percent of TSC patients develop kidney tumors that typically impair function, and may also cause severe bleeding.
Mutations in the TSC1 or TSC2 gene can cause the disease. However, those mutations are not usually inherited, and tend to happen during development or in early life. TSC tumors are often heterogeneous, even in the same patient. As such, animal models or treatments have been difficult to develop.
For this work, which was published in Cell Reports, the researchers created a new model, noted first study author and MD/PhD student Adam Pietrobon of the University of Ottawa. They genetically engineered kidney organoids using cells from human tissue.
Human stem cells were genetically reprogrammed to function as kidney cells that also carried mutations in TSC1 or TSC2. Individual cells were isolated from the organoids, then injected into a mouse model. Inside the mice, human TSC tumors grew.
This work showed that TSC tumors start with Schwann cell precursors, and that the TSC mutations impact many types of cells. This illustrates how the heterogeneity arises.
“The cells at the origin of tuberous sclerosis tumours have been a mystery for decades,” said senior author Dr. Bill Stanford, senior scientist at The Ottawa Hospital and professor at the University of Ottawa. “Our results can help find possible treatment targets for this challenging disease.”
Reporting in Cell Stem Cell, a different team of researchers gained new insights into autosomal dominant polycystic kidney disease (ADPKD), which affects about 8 million people around the world. The disorder, which is caused by mutations in either the PKD1 or PKD2 genes, causes cysts to form in the kidney. These fluid-filled sacs disrupt kidney function, and can lead to other complications that impact organs like the heart, liver, and pancreas. One approved drug for PKD only treats certain cases and does not halt disease progression.
In this study, pluripotent stem cells were genetically reprogrammed to grow into the filtering portion of the kidney, known as nephrons. Developing these organoids was a straightforward process. CRISPR/Cas9 tools were also used to introduce the PKD mutations into the kidney nephron organoids. "Most importantly, the organoids can consistently recapitulate key aspects of normal human kidney development, as well as cyst formation in ADPKD," noted corresponding study author Andy McMahon, a Professor and Chair at the University of Southern California (USC).
The organoids were used to perform a drug screen to identify new therapeutics that can treat ADPKD.
In the screen, the researchers focused on 247 enzyme inhibitors that could target the cellular mechanisms that may control cyst formation. Nine of those compounds successfully halted cyst growth, and did not appear to harm the organoids. While further research will be needed, a candidate called quinazoline, was found to be especially effective.
Sources: The Ottawa Hospital, Keck School of Medicine of USC, Cell Reports, Cell Stem Cell