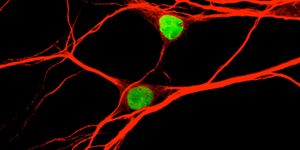
A team led by scientists at The Scripps Research Institute (TSRI), La Jolla, Calif, has determined the basic structural organization of a molecular motor that hauls cargoes and performs other critical functions within cells.
Biologists have long wanted to know how this molecular motor-called the "dynein-dynactin complex"-works. But the complex's large size, myriad subunits and high flexibility have until now restricted structural studies to small pieces of the whole.

In the new research, however, TSRI biologist Gabriel C. Lander, PhD, and his lab, in collaboration with Trina A. Schroer, PhD, and her group at Johns Hopkins University, created a picture of the whole dynein-dynactin structure.
"This work gives us critical insights into the regulation of the dynein motor and establishes a structural framework for understanding why defects in this system have been linked to diseases such as Huntington's, Parkinson's, and Alzheimer's," Lander says.
The findings are reported in a Nature Structural & Molecular Biology advance online publication on March 9.
The proteins dynein and dynactin normally work together on microtubules for cellular activities such as cell division and intracellular transport of critical cargo such as mitochondria and mRNA. The complex also plays a key role in neuronal development and repair, and problems with the dynein-dynactin motor system have been found in brain diseases including Alzheimer's, Parkinson's, and Huntington's diseases, and amyotrophic lateral sclerosis (ALS). In addition, some viruses (including herpes, rabies and HIV) appear to hijack the dynein-dynactin transport system to get deep inside cells.
The structural data clarify how dynein and dynactin fit together on a microtubule, how they recruit cargoes, and how they manage to move those cargoes consistently in a single direction.
[Source: TSRI]