The mechanisms that underlie physiological processes involve complex relationships between molecules. The proteins of a cell must work together to accomplish the tasks that are essential to life. That led researchers to look for the proteins that interact with the enzyme Protein phosphatase 2 (PP2A), which has a wide variety of roles in a cell but has been specifically has been linked to cancer growth.
A new publication in the journal Structure reports 98 proteins that are very likely to bind to PP2A. Those proteins are all now candidates to further study when searching for the reason why the pathways PP2A is linked to cause cancer when they are not functioning properly.
The study, by an international team of scientists at Brown University and University of Leuven in Belgium, investigated the three dimensional structure of the binding of PP2A with other proteins to find the proteins that would fit best. The majority of the proteins found in this work are not yet well understood, or even known.
Data obtained in the study may also be useful for predicting the binding strength between PP2A and one of the potential partners or substrates. Another use might be to look for ways to prevent a particular interaction, if it should turn out to be disease-causing in certain cases.
"PP2A is a very important enzyme, but identifying its substrates and its regulators has been exceptionally difficult and part of the reason is we didn't understand how they were engaging with this protein," commented the corresponding author of the publication, Rebecca Page, a Professor of Molecular Biology, Cellular Biology and Biochemistry at Brown University.
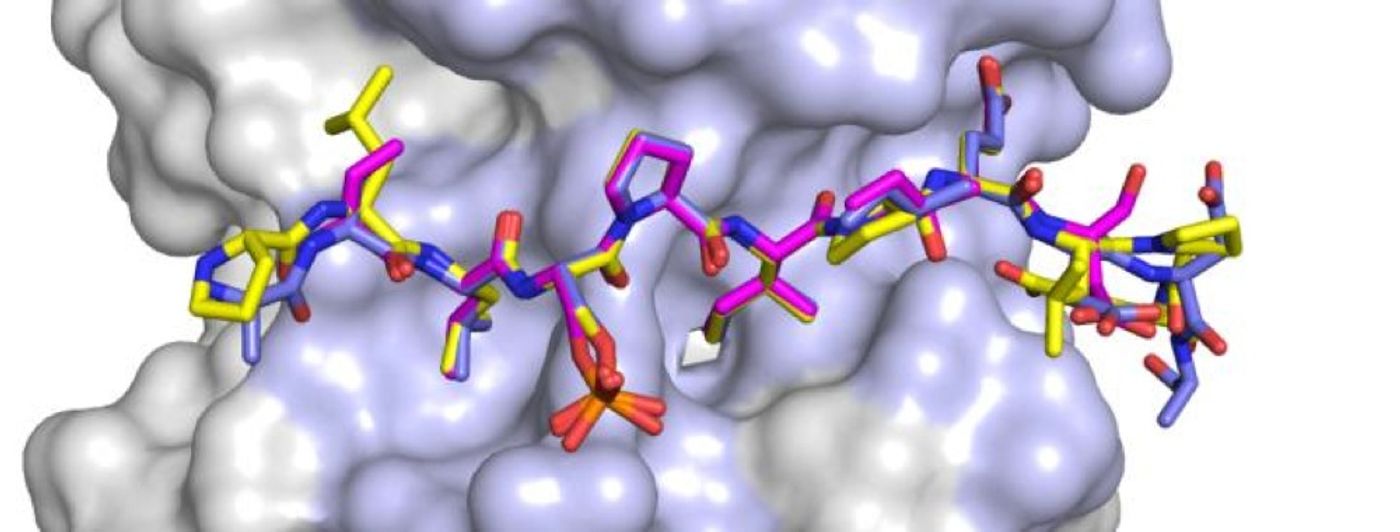
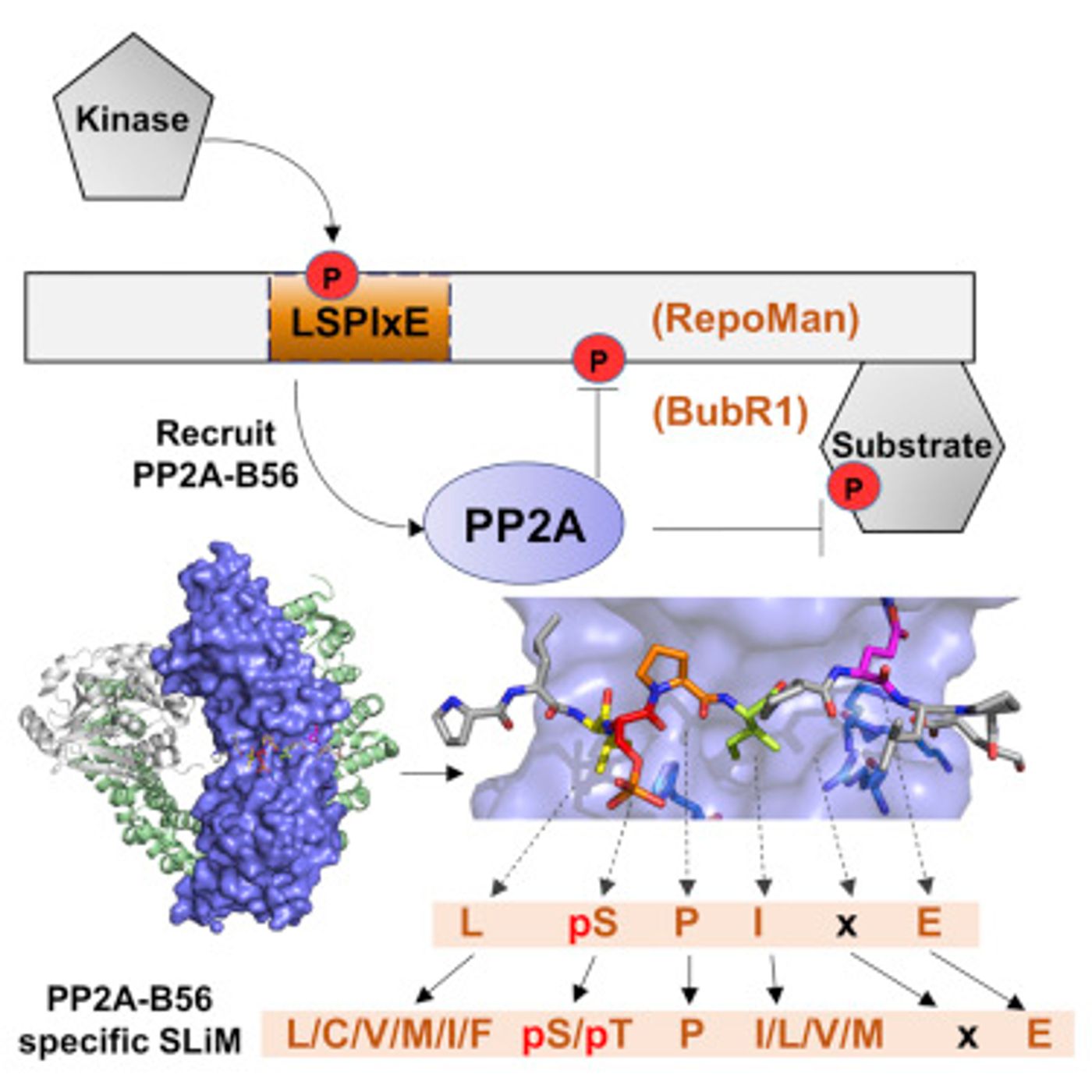
The effort to determine the best candidate substrates for PP2A was spearheaded by Xinru Wang, a Brown graduate student and Rakhi Bajaj, a postdoctoral researcher. They used shortened versions of two proteins that are already known to be associated with PP2A, BubR1 and RepoMan, and the regulatory region of the PP2A molecule. The resulting protein complexes were then made into crystals, enabling atomic-level analysis with X-rays. Their procedure allowed them to closely analyze the binding interactions and find consistencies and specialties.
"The similarities were the common features that we used to identify additional PP2A interactors," Wang explained. Page added, "The differences between RepoMan and BubR1 was what allowed us to explain why the binding affinities were different and how different PP2A regulators might be competing in the cell."
They used their data and observations from the crystallized proteins to scour databases of human proteins. Amino acid sequences gave them clues into the three dimensional structures that would likely bind PP2A. The 98 potential interactors were found after applying strict parameters; by looking for only the most likely partners of PP2A. Some of the proteins turned out to be known to interact, which validates their approach. Many others, however, are new.
"These new proteins provide targets that we can now go investigate in detail in cells to see what consequences changing these sequences have on their activity," Page said.
Bajaj noted that their data could already have had an impact on cancer research. For example, there is a a mutation causing a change in an amino acid, specifically Histidine 243, on PP2A-B56 that is found in germ cell tumors. The new research may help elucidate why. The team observed that the same amino acid plays an important role in PP2A’s binding process.
Sources:
AAAS/Eurekalert! via
Brown University,
Structure