Aging in Mice Slowed Through Cellular Reprogramming
Reporting in Cell, scientists were able to extend the lifespan of mice that carry a genetic mutation that causes premature aging. The scientists reprogrammed the epigenetic tags carried in the genome of the mouse, which both reduced many symptoms of aging and increased their lifespan from 18 to 24 weeks on average. The results of this work suggest that epigenetics exerts an influence on the aging process that might be changeable. The work is discussed in the following video.
"We did not correct the mutation that causes premature aging in these mice," explained lead investigator Juan Carlos Izpisua Belmonte, a Professor at the Salk Institute of Biological Science's Gene Expression Laboratory. "We altered aging by changing the epigenome, suggesting that aging is a plastic process."
While there have been attempts to tinker with lifespan in mice before, the result caused extensive tumor development or sudden death. In this breakthrough work in a live animal, the Salk research team performed a partial reprogramming of cells. Cellular reprogramming makes an adult cell turn into an induced pluripotent stem cell – a kind of cell that has the power to become any kind of cell in the body. The reprogramming process involves expressing four transcription factors, proteins that cause the expression of other genes; in the case of iPSCs they are referred to as Yamanaka factors for the scientist Shinya Yamanaka, who won a Nobel for his work in developing this technique.
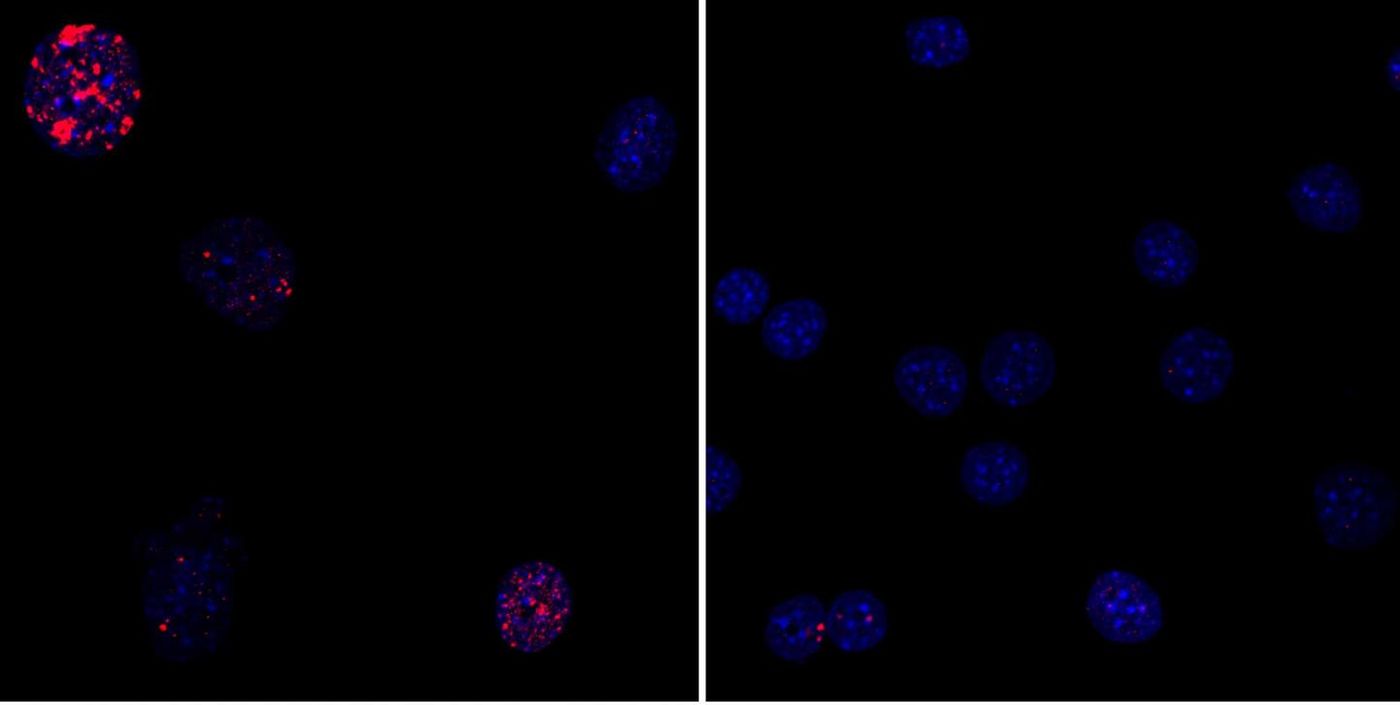
The researchers in this work wanted only a partial reprogramming; rather than inducing the expression of Yamanaka factors for the two to three weeks that is typical, they instead only went for two to four days. The cells did not reach a state of pluripotency as in iPSCs and remained specialized. However, dysfunction caused by aging was reduced in cells; there was a restoration of nuclear structure and less cumulative DNA damage. "These changes are the result of epigenetic remodeling in the cell," said Izpisua Belmonte. Tumors and death did not result from this method, only a lengthening of lifespan.
"We were surprised and excited to see that we were able to prolong the lifespan by in vivo reprogramming," said the co-first author of the study, Pradeep Reddy.
Throughout life, environmental influences have an effect on the epigenome, which effects genetic expression in turn. "During aging, marks are added, removed, and modified," explained co-first author Alejandro Ocampo. "It's clear that the epigenome is changing as we get older."
The partial induction of Yamanaka factors occurred in the entire organisms, and several organs were improved as a result. The appearance of tissue from skin, stomach, spleen and kidney was improved, and the cardiovascular system - a common failure in aged mice - had a better structure and function. "It is difficult to say specifically why the animal lives longer," said co-first author Paloma Martinez-Redondo. "But we know that the expression of these factors is inducing changes in the epigenome, and those are leading to benefits at the cellular and organismal level."
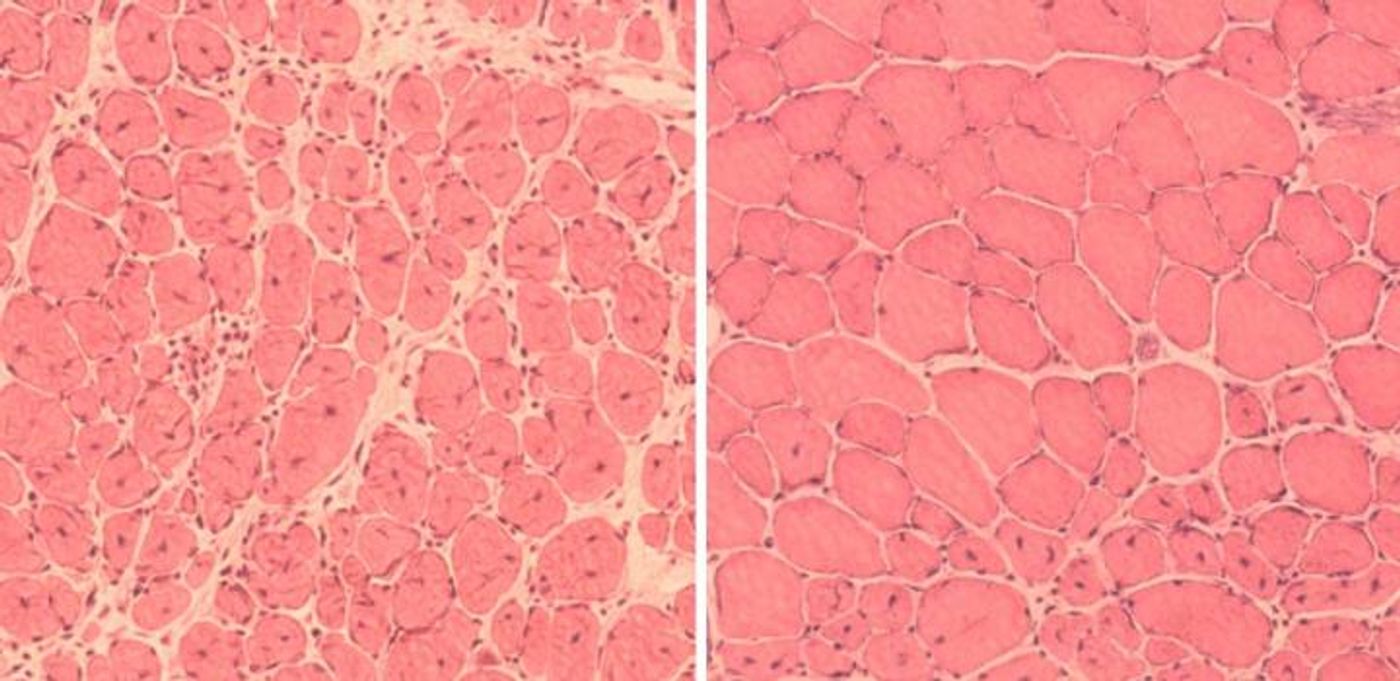
The team also wanted to see how this technique looked when applied to an injury. In a mouse model, muscle tissue and beta cells of the pancreas showed enhanced regeneration from the partial cellular reprogramming technique.
The researchers would like to continue this work to look at exactly what happens to the epigenome. "We need to go back and explore which marks are changing and driving the aging process," concluded Izpisua Belmonte.
Sources: AAAS/Eurekalert! via Cell Press, Cell