Synthetic Molecules Created for Future Therapeutic Potential
Chemists working at the University of Oregon have created a new kind of molecule with the aim that when broken down by the body under specific conditions, the designer molecule would induce the release of hydrogen sulfide. That could bring therapeutics to the cellular level, aiming to repair cells damaged by stress such as that from a heart attack. The work is far from use in the clinic, but has potential for the future.
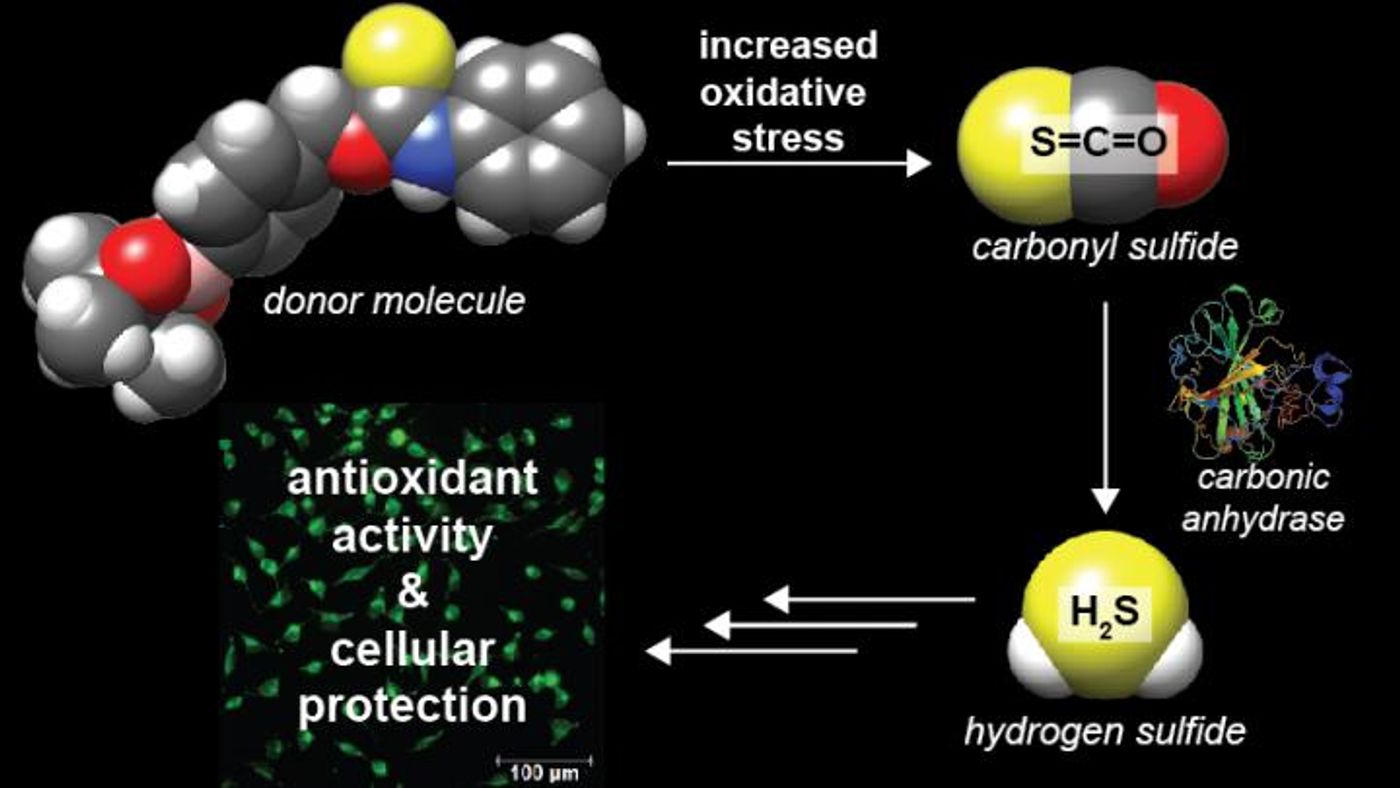
The proof-of-concept work was published in portions in the Journal of the American Chemical Society last June, and in November in Angewandte Chemie. "We have discovered that small organic molecules can be engineered to release a molecule called carbonyl sulfide, which is the most prevalent sulfur-containing molecule in the atmosphere, but more importantly converts rapidly to hydrogen sulfide under biological conditions," explained a co-author on both reports, Michael Pluth, a Professor of Chemistry. "We developed and demonstrated a new mechanism to release small molecules that provide therapeutic hydrogen sulfide."
Hydrogen sulfide doesn’t spring to mind when considering therapies, and is known as a dangerous and toxic colorless gas in the environment. However, hydrogen sulfide is also made by mammals, including humans, and plays critical roles in cardiac health and molecular signaling. As such, figuring out how to get the body to produce restorative hydrogen sulfide has been a goal for many researchers for the past two decades. The Pluth lab has previously invented a probe that is able to detect the gas in biological samples, setting up a way to assay candidate donor molecules – either those purified from natural sources or synthesized in the lab.
In the event of a heart attack or blood flow constriction, levels of reactive oxygen species like hydrogen peroxide increase, Pluth said. The newly created molecules are able to react to an increased expression of reactive oxygen species. Hydrogen sulfide donors that are available tend to be molecules that act slowly, only donating hydrogen sulfide in a passive way.
These two studies together indicate that molecules can be engineered to release carbonyl sulfide and then hydrogen sulfide by putting a trigger into the molecule that starts the delivery process, Zhao said. "With this, you might be able to pick molecular events that are associated with conditions in which hydrogen sulfide might be beneficial, and then develop donor molecules able to deliver hydrogen sulfide under those conditions," he explained.
"The novelty for us was being able to use carbonyl sulfide as a source of hydrogen sulfide donation," commented Steiger, regarding the project she led. "This was a first. It opened up a whole new class of donor molecules."
The development of these hydrogen sulfide-releasing molecules is a goal because there is real potential for eventual therapeutic applications, Pluth said. "Having researchers nearby who are focused on translating basic science discoveries into market applications would facilitate further expansion of this work," he concluded.
As previously mentioned, hydrogen sulfide has been on the research radar for awhile. A report is described in the cheeky video above in which a doctor at the University of Exeter identified hydrogen sulfide, a gas in flatulence, as a possible cancer therapeutic.
Sources: AAAS/Eurekalert! via University of Oregon News, Journal of the American Chemical Society, Angewandte Chemie