Photodynamic therapy is a cancer treatment that uses certain wavelengths of light to activate photosensitive drugs that are injected into the body. The photosensitive drugs then release free oxygen that kills the tumor cells. Photodynamic therapy can be applied to cancers that are relatively shallow in the body but the challenge remains with deep tissue tumors such as those in the lung or colon. University of Massachussetts Medical School scientist Gang Han, PhD, and his team have developed a new technique to allow photodynamic therapy to be effective on deeper tissue tumors.
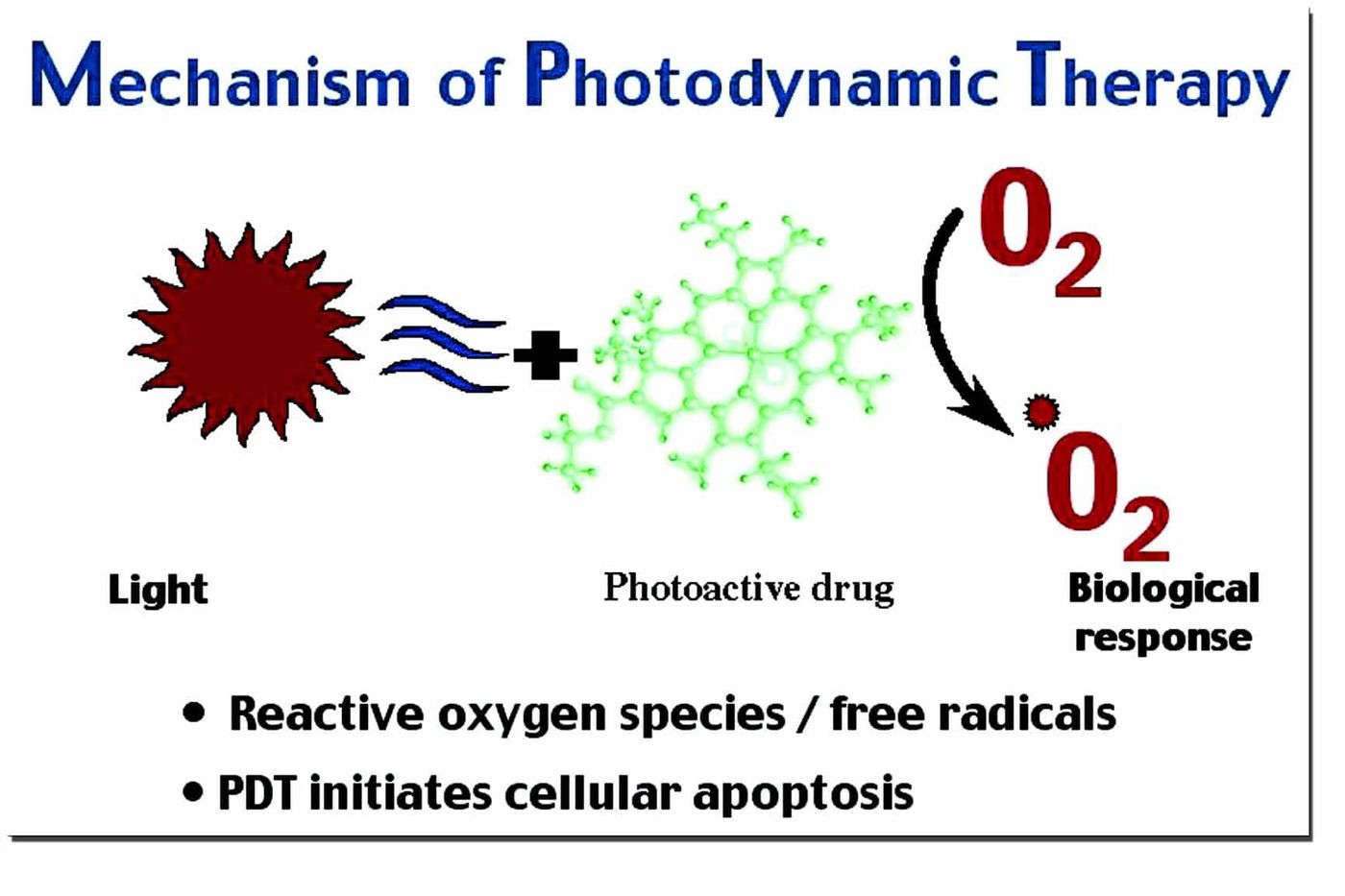
Dr. Han developed a near infrared (NIR)-light-absorbing carbazole-substituted BODIPY (Car-BDP) molecule that is suitable for use in the body and is highly effective as a deep-tissue photosensitizer for photodynamic therapy. Near infrared light absorption ranges from 600-800nm, so the molecule can be activated by a low powered and more cost-effective lamp light. Typical photodynamic therapies use a red laser light to activate the molecules. These molecules are also capable of holding a high density of singlet oxygen molecules for release upon light activation.
The Car-BDP molecules are encapsulated with specialized biodegradable polymers, enabling the Car-BDP to create uniform organic nanoparticles. The resulting nanoparticles can be mixed into solution to be injected into the patient. Instead of allowing the molecules to be absorbed into every cell in the body, then waiting for them to leave healthy cells and remain in tumor cells for treatment as is typical for this therapy, these molecules are tumor-targetable. The molecules can travel through the body, target deep tissue tumors, and outline and kill them upon activation.
“This study signals a major step forward in photodynamic therapy by developing a new class of NIR-absorbing biodegradable organic nanoparticles for a highly effective targeting and treatment of deep-tissue tumors,” Han said.
In the study, published in the
Journal of the American Chemical Society, the team tested the longevity of the nanoparticles by tracking the molecules using their NIR fluorescence capabilities as they travelled in vivo. As expected, the molecules are able to survive a long time travelling through the body without degradation. In addition, the molecules can be removed from the body, which is important for therapy drug development.
The nanoparticles signal an important advancement in the development of practical use photodynamic therapy. Han and his team are working toward creating this as a new platform for affordable targeted cancer treatments.
Sources:
UMass Medical School News,
ACS