Five Questions to Help You Select the Best Cellular Stain
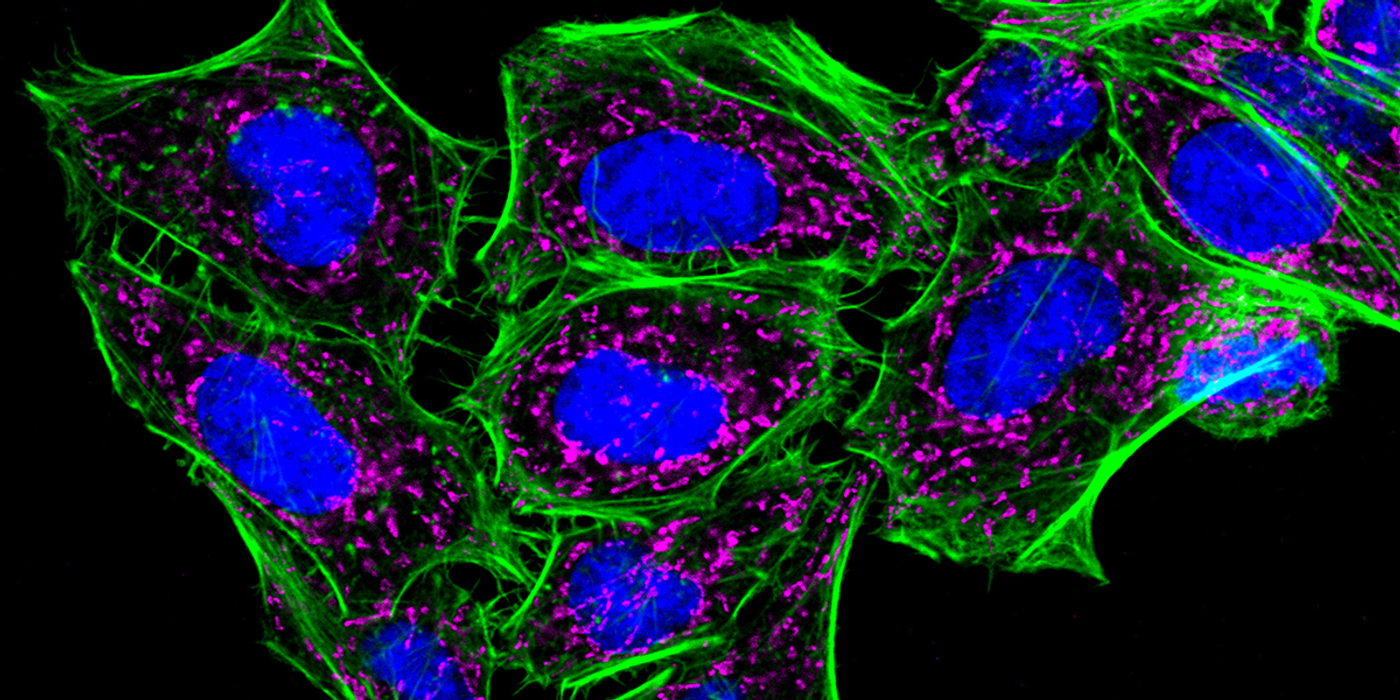
Cellular stains are organic fluorescent dyes or fluorescent conjugates designed to localize to a specific organelle or cellular structure. In some cases, the stains respond to the physiological environment, such as membrane potential or pH. They are widely used in fluorescence microscopy and flow cytometry to study cellular health and organelle function. Selecting the right stain is critical to getting meaningful results, and there are several things to keep in mind when making a selection.
An important consideration is whether the dye's fluorescence spectral properties are compatible with your instrument's excitation source and emission filters. In addition, photostability is an essential dye characteristic for fluorescence microscopy, while choosing brighter dyes is generally beneficial for flow cytometry. Finally, consider the experimental workflow, including downstream treatments after staining. Here are some questions to ask when selecting a cell stain that fits your workflow:
Is the stain cell type specific?
Most cellular stains will work for mammalian cells and other stable cell culture lines like insect cells. However, some stains bind to specific proteins and are thus dependant on protein expression levels. In this case, each cell line will have a different staining pattern or even none at all. In addition, not all cell stains work for bacteria or yeast. For instance, Wheat Germ Agglutinin (WGA) conjugated to dyes, such as the CF® Dyes, can stain cell surface outlines in many immortalized cultured cell lines. However, in tissue sections its localization is highly tissue- and cell-type specific, and will vary depending on the expression of cell surface glycoproteins. WGA conjugates also will stain gram-positive bacteria as well as yeast. On the other hand, a stain like Calcofluor White is specific to chitin, which is only found on the cell surface of fungi and gram-positive bacteria and not in mammalian cells.
Will the dye be used to stain live cells or fixed cells?
In live cells, the dye must permeate the plasma membrane, whereas this is not a consideration when using fixed cells. When working with fixed tissue, we need to keep in mind that treatment with formaldehyde or alcohol alters the details of some structures in the cell. For instance, preparing formalin-fixed paraffin-embedded (FFPE) tissue for imaging removes much of the membrane lipids. Consequently, lipophilic dyes that stain the cell surface by binding the plasma membrane aren't appropriate for staining FFPE tissue sections.
Will live cells be stained and monitored for extended periods?
Imaging live cells makes it possible to study functional changes in a cell over time. However, this poses a different set of demands on the dyes than when using fixed cells. The conditions during imaging should maintain cell viability throughout the observation, so dyes need to be non-toxic and stably label the structure of interest over time.
Can the dye be fixed after staining?
If you plan to fix the cells after staining, but before imaging or collecting data, the dye needs to withstand the chemical fixation and remain specific to the organelle even after the cells are dead. Some cell stains will transfer to other structures, while others, such as mitochondrial stains, will become unresponsive when the organelle is no longer functional. For example, MitoView™ 633 localizes to mitochondria with healthy membrane potential in live cells. However, staining is lost when fixing the cells because the mitochondrial membrane potential is depolarized. To create a mitochondrial dye compatible with downstream workflows like immunofluorescence, scientists at Biotium developed MitoView™ Fix 640, which is initially potential-dependant during live-cell staining but reacts covalently with mitochondrial proteins so that it is retained after fixation and permeabilization.
Will downstream treatments such as detergent permeabilization affect the staining?
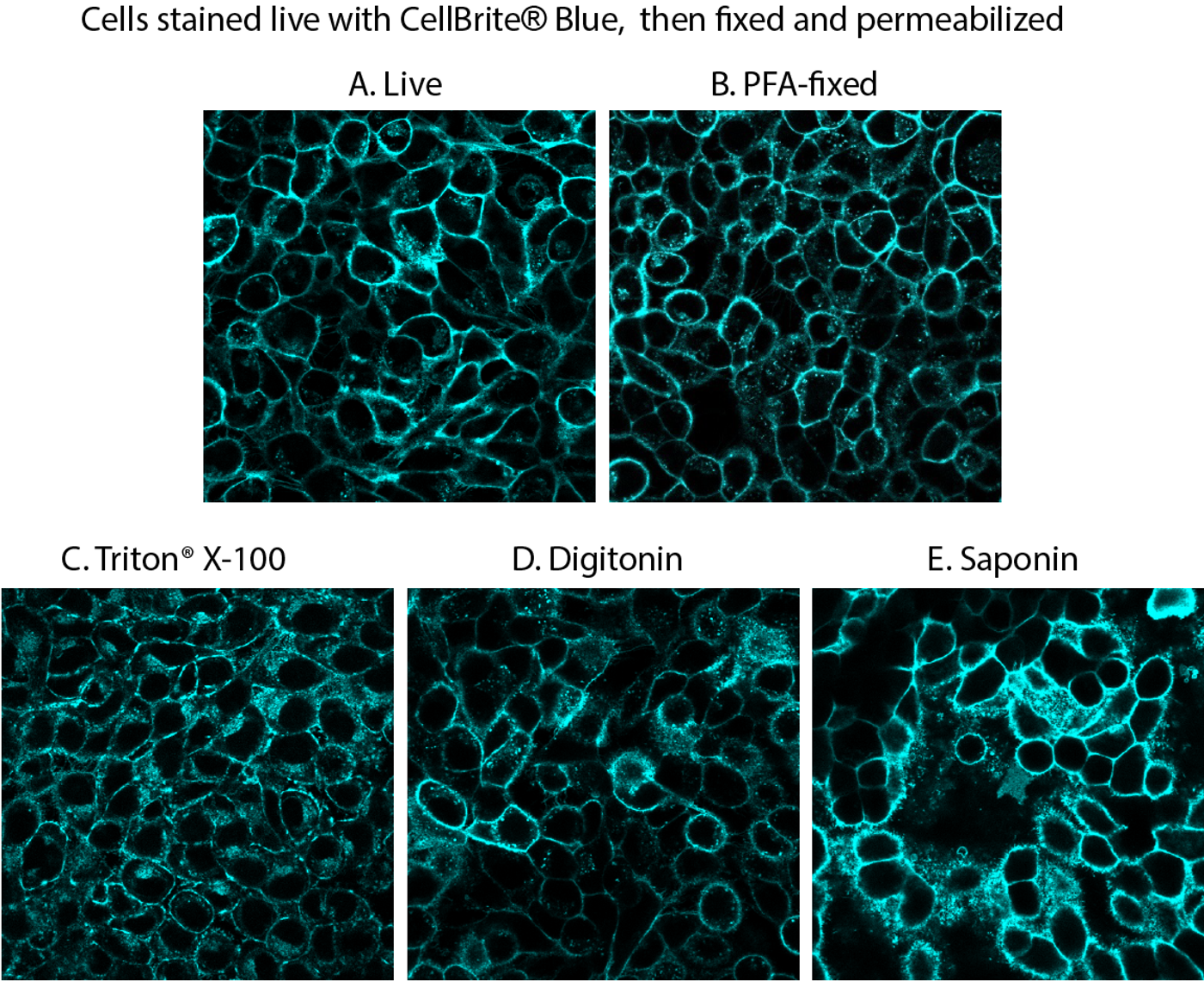
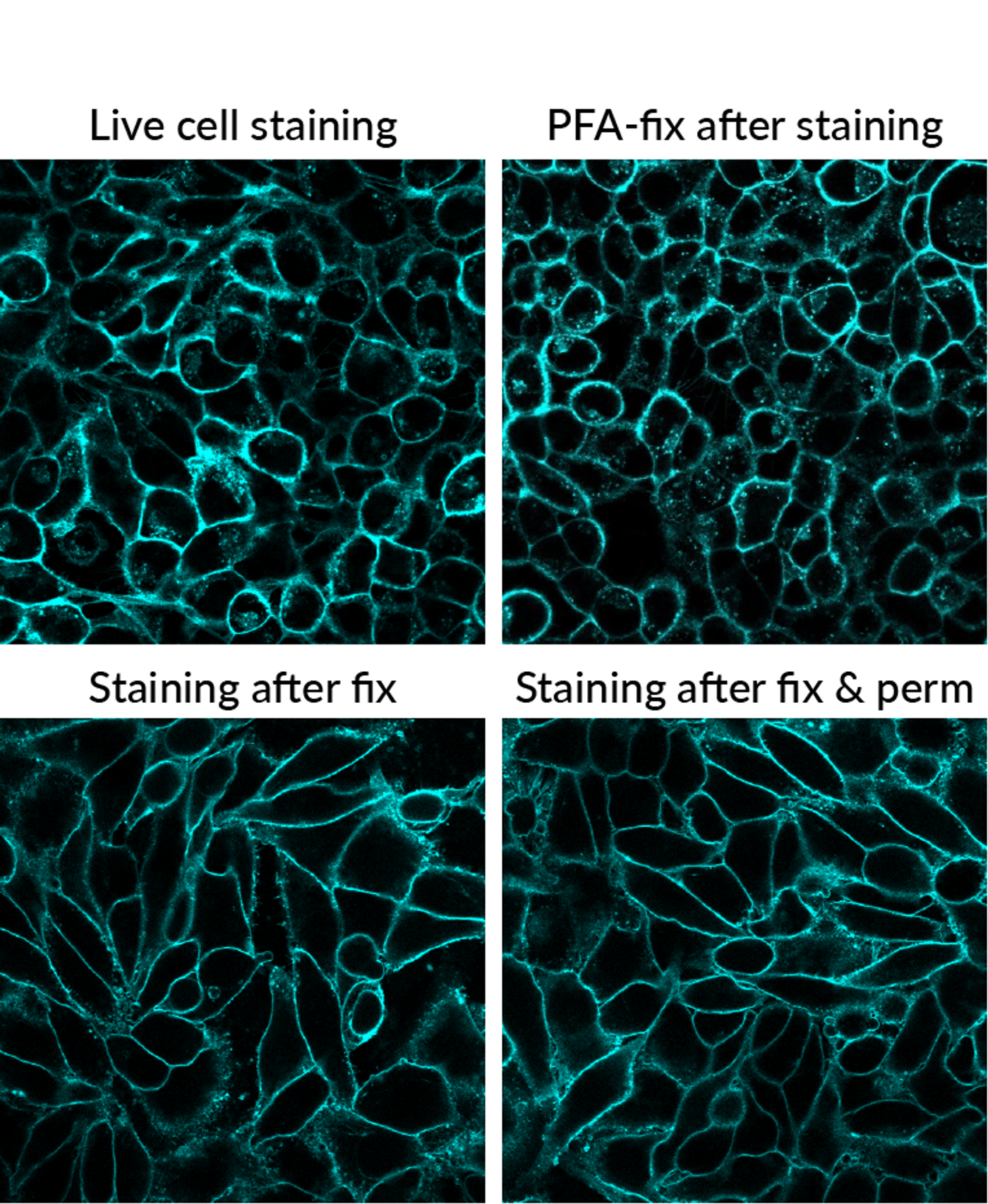
While stains that can be fixed will usually also withstand permeabilization with detergent, this is not always true. It is essential to consider the compatibility of the stain with downstream treatment of the sample in your workflow. For instance, using detergents such as Triton® X-100 or digitonin to permeabilize cells after staining with the CellBrite® Cytoplasmic Membrane Dyes will perturb the staining (See Fig. 1). Instead, CellBrite® Fix or MemBrite® Fix should be used because these dyes will tolerate these experimental procedures. Conversely, specific staining of cell surface membranes is better preserved when the cells are stained after fixing and permeabilization (Fig. 2).
Figure 1. Comparison of fixation/permeabilization protocols for CellBrite® Blue staining. Live HeLa cells were stained with CellBrite® Blue, then fixed with paraformaldehyde (PFA), followed by permeabilization with the indicated detergent at 0.1% in PBS for 10 minutes at room temperature. Cell surface staining was well-preserved after fixation (B), but intracellular staining increased after permeabilization with commonly used detergents (C-E).
Figure 2. Comparison of staining pattern with CellBrite® Blue under different workflow conditions. Find the protocol for combining cell surface staining with fixation and permeabilization for immunofluorescence in our tech tip.
If it seems impossible to sort through all of the dye characteristics you are not alone. That’s why Biotium has created the following resources to help you find the right dye for your experiment:
Cellular Stains Technology Page, listing Biotium's stains for different organelles.
Cellular Stains Selection Guide and Brochure, listing stains for different workflows.
Membrane and Cell Surface Stains Selection Guide and Brochure
Comparison of Cellular Stains in Different Organisms
At Biotium, the chemists who create the dyes collaborate closely with the biologists who develop and test products. In addition, our tech support scientists communicate with both teams, so the customer experience becomes a part of the development process. Together we are constantly developing new cellular stains with improved signal, higher specificity, more color selection, and better workflow compatibility.