Diagnosing and Treating An Extremely Rare Genetic Disease: Progeria
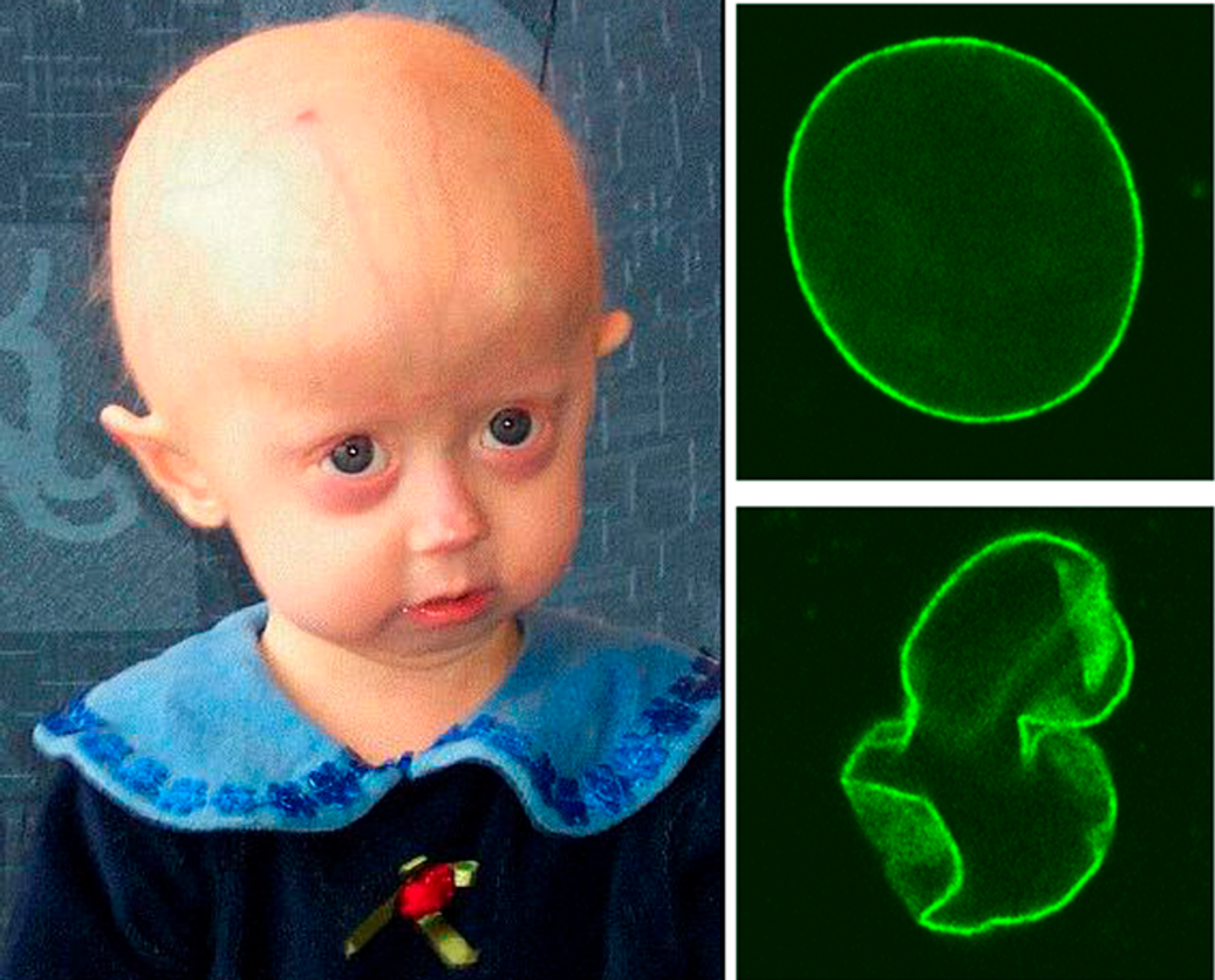
An extremely rare genetic disease that causes accelerated aging and heart disease has been little studied, largely due to a lack of relevant animal models. But now, scientists from the Centro Nacional de Investigaciones Cardiovasculares introduce the first mice genetically modified to illustrate atherosclerosis in the context of this rare disease, progeria, also known as Hutchinson-Gilford syndrome (HGPS).
Less than 400 people in the entire world have progeria. The disease is caused by a mutation in the LMNA gene, which produces a protein called Lamin A. This is a protein responsible for providing a foundation for holding a cell’s nucleus together. When Lamin A production is disrupted, the nucleus becomes unstable, which leads to premature aging characteristic of progeria. There is no effective treatment for this rare disease.
In addition to losing hair and body fat, osteoporosis, joint stiffness and wrinkled skin, progeria is characterized by atherosclerosis, a form of heart disease where plaques develop in the blood vessels and prevent healthy blood flow. When plaques rupture, heart attack or stroke can occur. Those affected by progeria rarely make it to their 15th birthday, and heart attack or stroke from ruptured atherosclerotic lesions is almost always the reason why.
But thanks to the new mice model of progeria, scientists can study the disease on a closer level than ever. For example researchers found that changes in the smooth muscle cell lining of blood vessels is largely responsible for atherosclerosis and premature aging in cases of progeria. Atherosclerosis is also triggered by progerin, a shortened version of the lamin A protein responsible for the disease. Cardiovascular deterioration and aging worsened by the accumulation of progerin.
"We found that smooth muscle cells in the arterial wall die early in the development of disease in the progeroid mice, and this induces an anomalous accumulation of lipoproteins in the vessels,” explained first author Dr. Magda Hamczyk. “This significantly increases the formation of atherosclerotic plaques, and the loss of smooth muscle also destabilizes the plaques and promotes plaque rupture in more advanced stages."
The new findings indicate smooth muscle cells from the lining of blood vessels as a possible therapeutic target for atherosclerosis in the context of progeria. Additionally, mice only expressed progerin in specific tissues linked to atherosclerosis, which might help scientists focus on where in the body progeria develops.
"This new animal model is allowing us to advance knowledge of the molecular and cellular mechanisms that cause cardiovascular disease and accelerated aging in progeria, an indispensable goal for the development of new therapies for patients with this severe disease," said Dr. Vicente Andrews. "Research into progeria can therefore help to identify mechanisms underlying normal aging and promote a healthier old age."
The present study was published in the journal Circulation.
Sources: Progeria Research Foundation, American Heart Association, Centro Nacional de Investigaciones Cardiovasculares