Rapidly Screening Genes in 'Living Test Tubes'
Rare diseases can be a terrible ordeal. Some symptoms may be obvious, others may be discreet, and diagnosing such disorders is often extremely challenging. One example is a seven-year-old boy that was born with no thymus, an organ that has important immune functions. He also had abnormal craniofacial features, heart and bone defects, and signs of autism. His mother and sister presented with similar, but milder defects. None of them carried gene deletions or alterations that were known to be associated with their abnormalities.
"His physicians thought he had many features commonly seen in children with 22q11.2 chromosomal deletion syndrome," said Dr. Shinya Yamamoto. Yamamoto was a co-corresponding author of a new report about a new method for studying rare diseases, and is an assistant professor of molecular and human genetics at Baylor College of Medicine and investigator at the Jan and Dan Duncan Neurological Research Institute at Texas Children's Hospital. After finding that none of the family members carried that genetic defect, researchers had to look elsewhere.
"This spurred Duke University investigators Dr. Vandana Shashi (featured in the video below) and Dr. Loren Pena and genetic counselor Kelly Schoch, who are a part of the Undiagnosed Disease Network, to perform whole-genome sequencing analyses on this family," Yamamoto said.
A rare missense mutation called p.R20Q in the TBX gene was found to be carried by all three family members that were affected. The team wanted to locate more people with the mutation, so they used GeneMatcher, a website that collects rare mutations and connects clinicians and researchers.
Another patient was found, at Cook Children's Hospital in Fort Worth, Texas. Many of the characteristics that were found in the first patient were also found in this individual, who actually carried a different mutation, p.R305H, in the TBX2 gene; that mutation was also a missense variant. TBX or T-box genes are known to be critical during development. Developmental syndromes have been traced back to errors in twelve of the seventeen members of the T-box family of genes.
An analysis of the TBX2 mutation revealed that it lowers levels of the TBX2 protein, which keeps it from carrying out its roles. This is the first connection between the TBX2 gene and an inherited disorder, a connection that is not always easy to make.
"Determining the functional consequences of missense mutations such as these - mutations that change a single amino acid in a protein - is still very difficult, even with the help of various bioinformatics tools," said Yamamoto.
The fruit fly was tested as a way to get a functional readout of gene mutations. The laboratories of Dr. Yamamoto, Dr. Michael Wangler, and Dr. Hugo Bellen, who directs the UDN's Model Organism Screening Center at Baylor, designed assays and tested them.
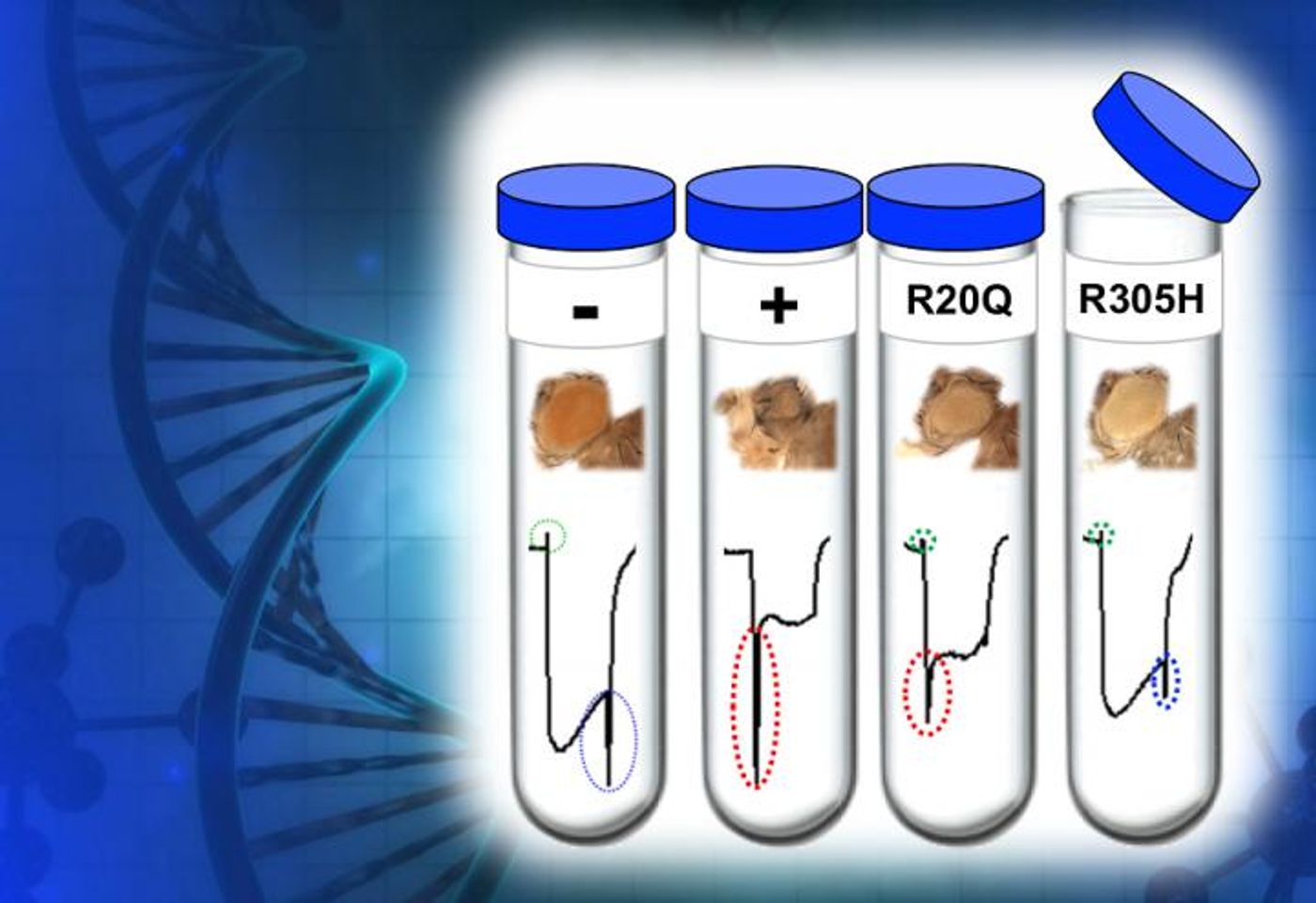
"We used fruit flies as 'living test tubes' in which we tested the effects of variations of the TBX2 gene on various biological processes and pathways," Yamamoto explained.
Animal models are a typical part of the research lab; the idea is study disease in a model that is very similar to the human system. The fruit fly, Drosophila, is one such organism. In this work, they enable a rare mutation to be rapidly assessed. Looking at TBX2 in the fly showed that the bone and heart defects, and immune and endocrine disorder seen in patients were indeed linked to the loss of function of the TBX2 protein.
"The human and fly circulatory and immune systems are structurally different, and flies do not have an endoskeleton system. Therefore, in this study, instead of using fruit flies to model the symptoms found in patients, we used flies as a rapid diagnostic tool to test whether the functional differences in TBX2 we found in cells in the laboratory had biological significance in a live animal," said Yamamoto. "The fruit fly's equivalent to the human TBX2 gene, called bifid, is important for the proper development of the fly, especially for the visual and nervous systems. We used the development and retinal function of adult fruit fly eyes as a readout for the activity of TBX2 gene."
The fruit fly test results confirmed what had been seen in cells growing in culture; TBX2 missense mutations impacted the eye, retina, and lifespan of the fly.
"This study provides a blueprint of how fruit flies can be used as a rapid screening tool to identify potentially pathogenic human genes," Yamamoto said.
Sources: AAAS/Eurekalert! via Baylor College of Medicine, Genome Biology, Human Molecular Genetics