Disruptions in Mitochondrial Quality Control Can Cause Heart Disease
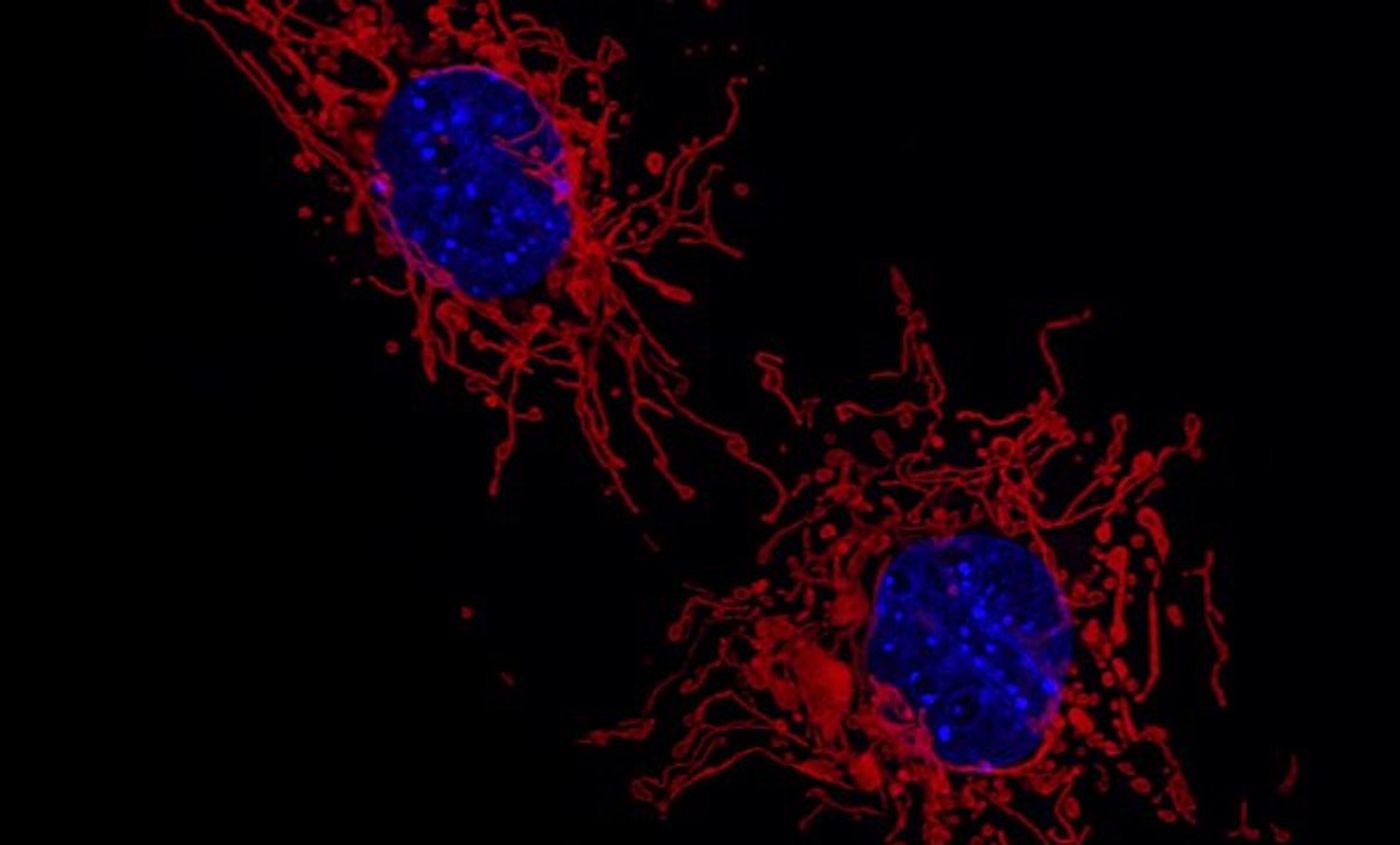
It’s known that when the ANT1 gene, which encodes for a protein called adenine nucleotide translocator (ANT) carries mutations, a variety of dysfunctions result, including eye muscle weakness and heart disease. Researchers at Penn Medicine have now learned more about why. Their findings, reported in Nature, showed that ANT is vital to a process called mitophagy, which helps remove damaged organelles named mitochondria, the powerhouses of the cell. Problems with this quality control mechanism can ultimately lead to heart defects.
ANT is well-known as a contributor to mitochondrial energy production, but this is a newly discovered function of the protein. Mutations in the ANT gene are linked to cardiomyopathy, which impairs the heart’s ability to pump blood, but these genetic errors don’t disrupt power production in the cell.
“In revealing the link between ANT and mitophagy, and the impact mutations have on quality control, our findings change the way we think about the disease-causing mutations and enable us to focus our attention on the right pathway,” said the corresponding author of the report, Zoltan Arany, M.D., Ph.D., a professor of Cardiovascular Medicine in the Perelman School of Medicine at the University of Pennsylvania. “Now that we know these diseases are caused by defective quality control rather than a lack of ATP generation, we can start thinking about therapeutic approaches that improve the quality control.”
Mitochondria help create the energy that cells need to carry out their functions, and are crucial to energy-intensive cells, such as those in the liver and heart. Mitophagy helps clear out defective mitochondria that have been impaired by stress, for example. This work has also revealed more about mitophagy, which is described in the video below.
In this work, the researchers used the CRISPR/Cas9 gene editor to show that ANT is necessary for mitophagy in various types of cells. They showed that ANT can promote mitophagy without influencing the production of cellular fuel, a molecule called ATP; the protein has two different functions.
Further research showed that mutations in ANT that cause disease impair the protein’s ability to bind to protein complexes that enable the movement of molecules from the inner membrane to the matrix of the mitochondria. If ANT is deleted, mitophagy is suppressed. When the gene is removed in a mouse model, mitophagy was reduced, and there was a buildup of unusual mitochondria.
“We were extremely surprised to find that ANT participates in mitophagy, revealing that it has two separate functions -- ATP generation and quality control,” Arany said. “While there’s no magic bullet for improving the quality control process, we hope our findings lay the groundwork for future efforts that examine how to offset the suppression of mitophagy and, ultimately, prevent these mutations from causing disease.”
Sources: Penn Medicine News, Nature