Unusual Mutation Acts as a Kind of Gene Therapy
Clinicians have identified a person with a rare inherited disorder that disrupts the production of fresh blood cells, a vital process, due to a mutation they carry in a gene called GATA2. However, the individual also carries another non-inherited or somatic mutation that prevents the disease from giving them symptoms.
GATA2 encodes for a molecule called GATA-binding protein 2, which helps control the activity of a variety of genes that are involved in development and cell renewal. Mutations in GATA2 disrupt blood cell replenishment, called hematopoiesis, and carriers experience symptoms like hearing loss, bone marrow failure, and lymphatic system blockage.
In the person identified in this study, a spontaneous mutation occurred early in their life after they began to develop, but it was not present from the beginning, or inherited like their GATA2 mutation was. This non-inherited or somatic mutation is believed to have had a corrective effect, and the patient did not have the standard symptoms of the disease. The findings, which could help other with the inherited disorder, have been reported in Blood.
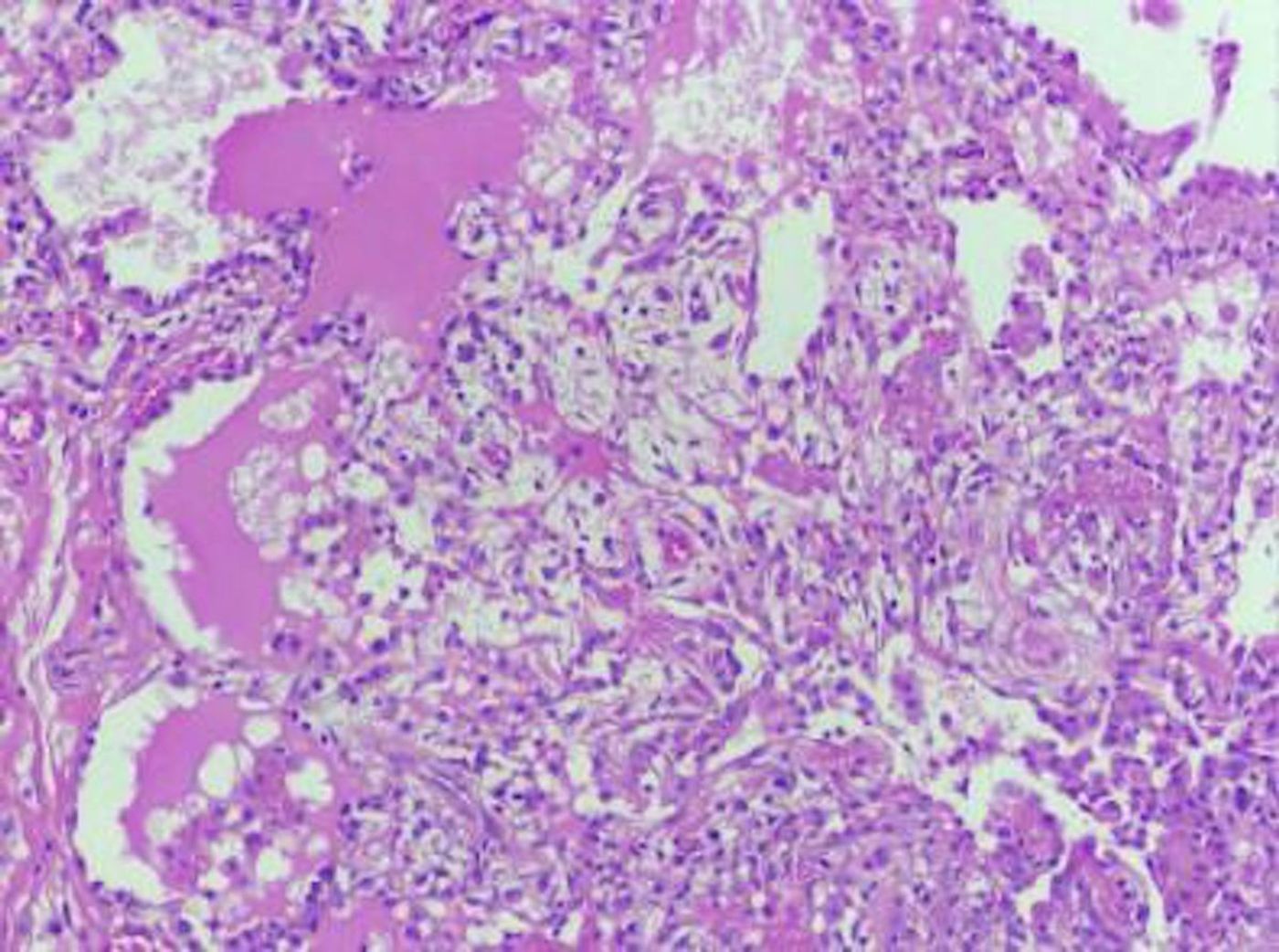
This study describes a family with two brothers that were receiving treatment for a bone marrow disease that disrupts blood cell production, called moderate aplastic anemia. One patient had a low red blood cell count and problems with their immune system for several years before dying at age 27 of a lung infection. The patient's brother had deafness, hypothyroidism, and recurrent lung infections. Both carried inherited GATA2 mutations.
"When a germline [inherited] mutation in GATA2 is detected, the patient's family has to be investigated because there may be silent cases," said the first study author Luiz Fernando Bazzo Catto.
The study authors looked to the DNA of the patients' parents to learn who had passed it down to them, and found that the mother did not carry the mutation. The father, however, had the same GATA2 mutation but was asymptomatic.
"This discovery raised the question whether the father transmitted the mutation or acquired it but didn't pass it on to his sons," Catto said.
Further investigations revealed that the father carried a somatic mutation in about 93 percent of his leukocytes, which was protecting him from the symptoms, while the other seven percent of his leukocytes were mutants that carried the disease-causing mutation in GATA2 but not the somatic mutation.
"This seven percent were a remnant of the original clone," Catto said. "It's very likely that the father had acquired the somatic mutation in his blood a long time ago."
An analysis of the telomeres in the peripheral blood leukocytes showed they were long, and telomeres get shorter as cells divide over and over.
"This indicates that these blood cells can remain active for a long time," Catto explained.
It may be that this somatic mutation is working to repair the problems with hematopoiesis that would otherwise occur due to the inherited mutation.
"A sort of natural gene therapy occurred in this patient," said corresponding study author Professor Rodrigo Calado of the University of São Paulo's Ribeirão Preto Medical School (FMRP-USP). "It's as if he embodied an experiment and a medium-term prospect of analogous gene therapy treatment in patients with GATA2 deficiency. The findings help us understand better how stem cells can recover by repairing an initial genetic defect."
Sources: AAAS/Eurekalert! via Fundação de Amparo à Pesquisa do Estado de São Paulo, Blood