Tumor Growth in NF1 Kids May Begin When Optic Nerve Fires
Young children who are born with neurofibromatosis type 1 (NF1) are at heightened risk of having vision problems because they are predisposed to optic nerve tumors. Scientists have now found that in these patients, the very act of seeing light might be causing tumors to form that disrupt vision. The optic nerve sends information from neurons in the eyes to the brain. The activity underlying this neurological process seems to trigger and promote tumor growth. In a mouse model, the researchers were also able to prevent the formation of these tumors by treating the mice with an experimental drug during a critical stage of development. The findings have been reported in Nature.
"Brain cancers recruit the resources they need from the environment they are in," said co-senior study author Michelle Monje, M.D., Ph.D., associate professor of neurology at Stanford University, Palo Alto, California. "To fight brain cancers, you have to know your enemies. We hope that understanding how brain tumors weaponize neural activity will ultimately help us save lives and reduce suffering for many patients and their loved ones."
Tumor cells don't grow in isolation, so scientists are beginning to investigate how tumors affect the environment around them and vice versa.
"Over the years, cancer researchers have become more and more focused on the role of the tumor microenvironment in cancer development and growth. Until recently, neuronal activity has not been considered, as most studies have focused on immune and vascular cell interactions," explained Jane Fountain, Ph.D., program director at the NIH's National Institute of Neurological Disorders and Stroke (NINDS). "This study is one of the first to show a definitive role for neurons in influencing tumor initiation. It's both scary and exciting to see that controlling neuronal activity can have such a profound influence on tumor growth."
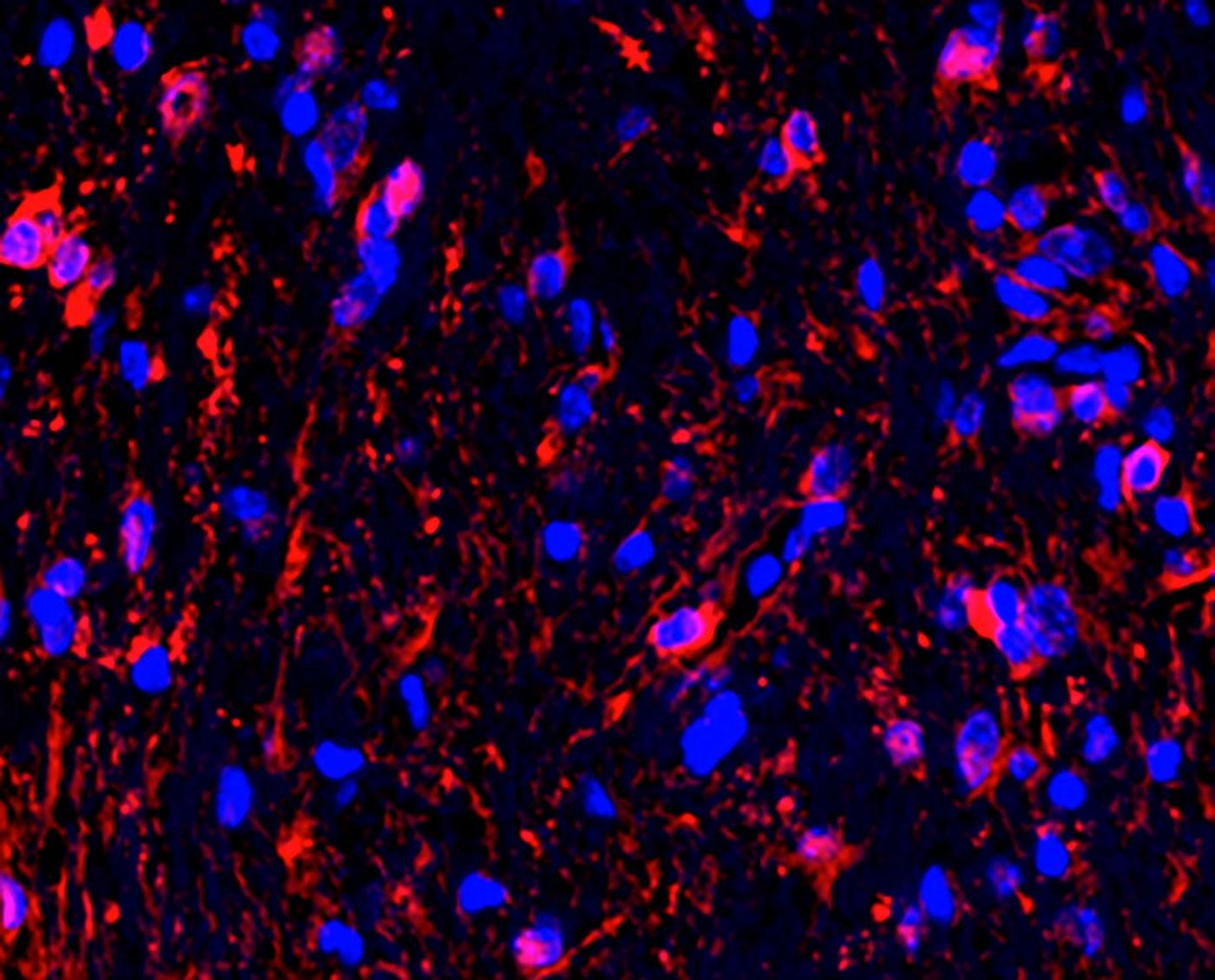
About 15 to 20 percent of kids that are born with a genetic mutation that causes NF1 go on to develop tumors, and in about half of that group, the vision is affected. Very similar tumors called optic nerve gliomas were used as a model in this research.
"Based on multiple lines of converging evidence, we knew that these optic nerve gliomas arose from neural precursor cells. However, the tumor cells required help from surrounding non-cancerous cells in the optic nerve to form gliomas," noted senior study author David H. Gutmann, M.D., Ph.D., the Donald O. Schnuck Family Professor and the director of the Neurofibromatosis Center at Washington University School of Medicine in St. Louis. "While we had previously shown that immune cells, like T-cells and microglia, provide growth factors essential for tumor growth, the big question was: 'What role did neurons and neural activity play in optic glioma initiation and progression?'"
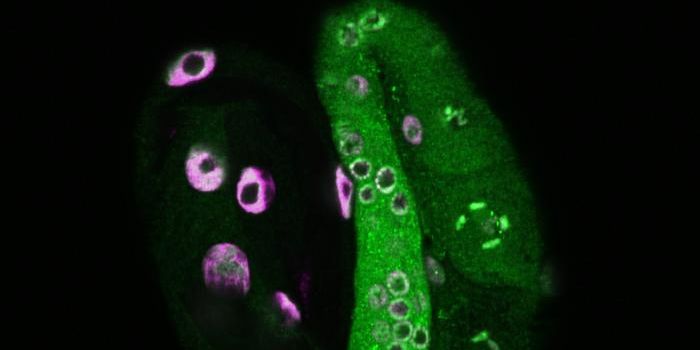
A mouse model of NF1-associated optic gliomas was used in this work; the mice typically develop these tumors between six and sixteen weeks of age. The researchers raised these young mice in different light conditions. Mice raised in total darkness during the six- to sixteen-week period did not develop tumors. Two of nine mice that were raised in the dark starting at twelve weeks of age did develop tumors, however, suggesting that timing is a factor.
"These results suggest there is a temporal window during childhood development when genetic susceptibility and visual system activity critically intersect. If a susceptible neural precursor cell receives the key signals at a vulnerable time, then it will become cancerous. Otherwise, no tumors form," said lead study author Yuan Pan, Ph.D., a post-doctoral fellow at Stanford. "We needed to understand how this happens at a molecular level."
Additional work revealed that a molecule called neuroligin-3 may be involved; when the gene was silenced in the mouse model, tumors did not form. Mice raised under different light conditions also had different levels of neuroligin-3 in their neurons.
"Previously our lab showed that neural activity causes shedding of neuroligin-3 and that this shedding hastens malignant brain tumor growth. Here our results suggest that neuroligin-3 shedding is the link between neural activity and optic nerve glioma formation. Visual activity causes shedding and shedding, in turn, transforms susceptible cells into gliomas," Monje explained.
In this comprehensive study, the researchers were also able to apply an experimental drug to prevent tumor growth. The drug is meant to reduce neuroligin-3 shedding. Treatment between six and sixteen weeks prevented tumor growth but treatment starting at twelve weeks did not prevent tumor formation; optic glioma growth was reduced, however.
Sources: AAAS/Eurekalert! via NIH/National Institute of Neurological Disorders and Stroke, Nature