With an enhanced form of the CRISPR-Cas9 system, scientists were able to read the genome of induced pluripotent stem cells (iPSCs) for the first time. The new system provides a more temporary gene modification that will be extremely useful for scientists using this method to conduct therapeutic gene editing techniques.
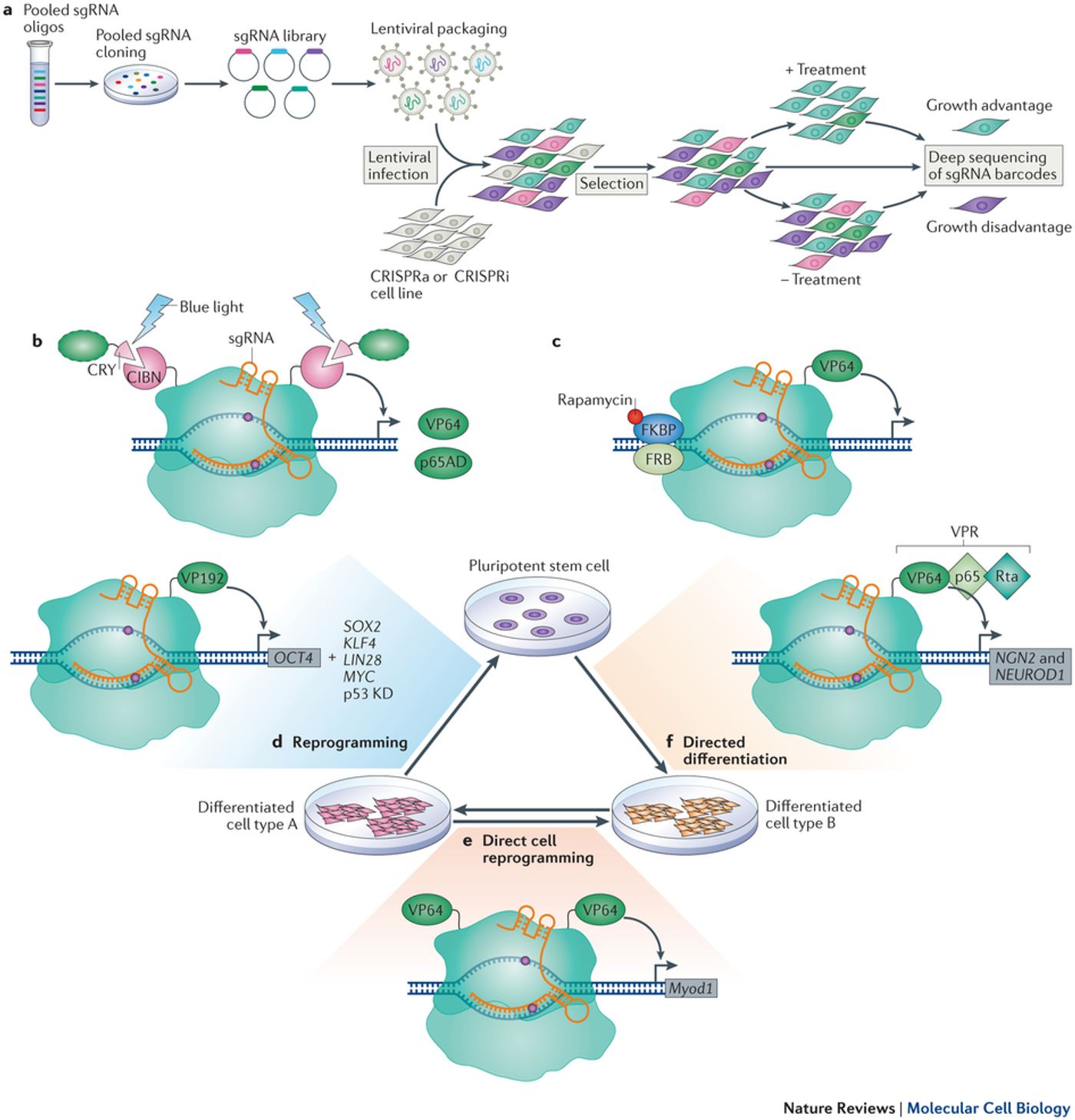
The new study builds off of a 2013 method developed by Stanly Qi, PhD, who is also a co-author on the current paper. Qi designed CRISPR interference (CRISPRi), a system with the ability to “inactivate genes in IPSCs and heart cells created from iPSCs.” CRISPRi improves the original system because genes can be silenced or turned off “more precisely and efficiently.” Plus, CRISPRi offers additional beneficial characteristics like deletion flexibility and reversible gene suppression.
Instead of the traditional Cas9 protein used by the original system, CRISPRi uses a deactivated version of Cas9 in addition to an inhibitor protein called KRAB. These proteins work together to suppress parts of the target genome without actually cutting the DNA like Cas9 does. Apparently this more temporary method of suppressing gene expression produces more consistent reads than when Cas9 makes permanent cuts in the DNA.
In the new Gladstone Institutes study published in Cell Stem Cell, scientists completed a comparison of traditional CRISPR-Cas9 and CRISPRi in their ability to silence an iPSC gene that controls pluripotency. While CRISPR-Cas9 was successful in silencing the target gene 60-70 percent of the time, CRISPRi appeared even more prominent, with a success rate of 95 percent.
"We were amazed by the dramatic difference in performance between the two systems," said senior author Bruce Conklin, MD. “In fact, CRISPRi is so precise and binds so tightly to the genome that it is actually a better way to silence a gene.”
In addition to being a reversible way to silence a gene, CRISPRi allowed the scientists to modify just how much a gene was silenced by adding different amounts of chemicals. This characteristic is especially important in clinical situations, since not all gene mutations completely suppress a gene’s expression.
"Using this technology, we can mimic disease in a homogenous population of heart cells created from iPSCs,” said first author of the study Mohammad Mandegar, DPhil. “This development allows us to study genetic diseases more easily and potentially identify new therapeutic targets."
Source:
Gladstone Institutes