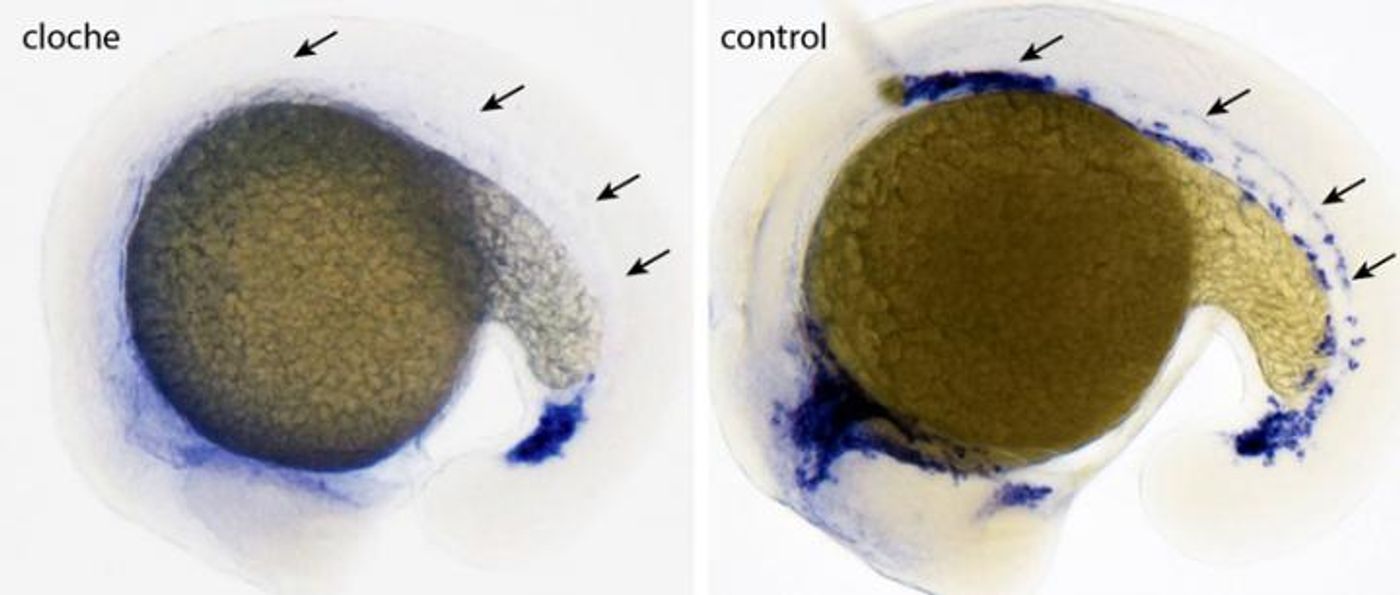
Over 20 years ago researcher Didier Stainier found a zebrafish mutant he named cloche, after comparing the shape of the mutant’s heart to that of a 1900s-style woman’s hat. The mutation present in the zebrafish severely impacted the development of both endothelial and hematopoietic lineages early on in embryonic development. Stainier couldn’t find a genetic culprit for cloche for a long time, but this week scientists have finally made a breakthrough.
Stainier, who is the director of the Max Planck Institute for Heart and Lung Research, noticed in his original 1995 study that cloche lacked normal development of both blood cells and blood vessels, a condition that was complicated by a selective deletion of the endocardium. While embryonic cardiac morphogenesis continued despite the missing endocardium, the growing cloche embryo saw an abnormal reduction in myocardial contractility due to the mutation.
The mutant hematopoietic tissues also showed zero expression of both GATA-1 and GATA-2, two important transcription factors whose expression is required during early embryogenesis to regulate genes during development and differentiation of hematopoietic cells. Finally, in a recent paper published in
Nature, Stainier and his team were able to finally uncover the gene responsible for the condition of the cloche zebrafish mutant.
They started by examining 26,000 genes from the cloche genome, all of which were cloche suspects. “Identifying cloche was like solving a decades-old criminal case of genetics,” said Sven Reischauer, one of the main authors of the study. “However, in this case, it was not the perpetrator who was unknown but the victim, the defective gene."
17 out of the 26,000 were then targeted as potential defective genes. To pinpoint the true mutant gene out of the lineup, the researchers created several knockout lines for each candidate, introducing them into zebrafish embryos, deactivating them, and then following blood vessel and blood cell growth in each subject to see if the condition matched that of cloche.
Finally, the researchers landed on one gene that was clearly responsible for the cloche, hidden all the way at the end of chromosome 13, a telomeric region. They continued to confirm this gene’s identify by conducting “overexpression experiments” where embryos received injections of pure cloche mRNA, after which scientists were able to prompt blood cell and blood vessel formation earlier than they normally occur in embryos.
With the cloche gene confirmed, researchers now ponder the many clinical applications that might arise from manipulating the expression of the gene. Of all the genes known to be related to blood vessel formation, they are all dependent on whether the cloche gene has been activated. Scientists believe this level of control could greatly contribute to personalized stem cell therapy and so much more.
Sources:
Max Planck Gesellschaft,
Development,
Nature