Using CRISPR For Epigenetic Modifications
CRISPR/Cas9 gene editing technology has been hailed as revolutionary, but the technique came with some caveats. The process involves making a break in both strands of double-stranded genomic DNA (DSB), and although the enzyme that cuts is directed to a particular region of the genome, it is not always as specific as intended. Therefore, its use in human patients is considered limited. Salk scientists have now refined the tool so it changes gene expression without making DSBs. Their method was used to successfully treat mouse models of several diseases, including muscular dystrophy, diabetes, and acute kidney disease.
"Although many studies have demonstrated that CRISPR/Cas9 can be applied as a powerful tool for gene therapy, there are growing concerns regarding unwanted mutations generated by the double-strand breaks through this technology," explained Juan Carlos Izpisua Belmonte, a professor in Salk's Gene Expression Laboratory. He is the senior author of a paper reporting their findings, published in Cell. "We were able to get around that concern."
Researchers have modified the Cas9 enzyme so that it will target a particular place in the genome, but without cutting it, a “dead” type of Cas9 (dCas9). This dCas9 was joined with molecular switches that turn gene expression on, transcriptional activation domains. That makes for a very large molecule, however. The delivery systems used for these therapies, adeno-associated viruses (AAVs) are not big enough to carry them into live organisms, presenting a significant hurdle for their use in the clinic.
The researchers instead used molecules that were not fused together. One batch of reagents - the transcriptional activators, and the guide RNAs - were put into one AAV; the dCas9 went into another. The researchers found that this worked to strongly activate a targeted gene.
"Basically, we used the modified guide RNA to bring a transcriptional activator to work together with the Cas9 and delivered that complex to the region of the genome we were interested in," explained co-first author Hsin-Kai Liao of the Belmonte laboratory.
"We wanted to change the cell fate with therapeutic efficiency without a DNA cut," said co-first author Fumiyuki Hatanaka.
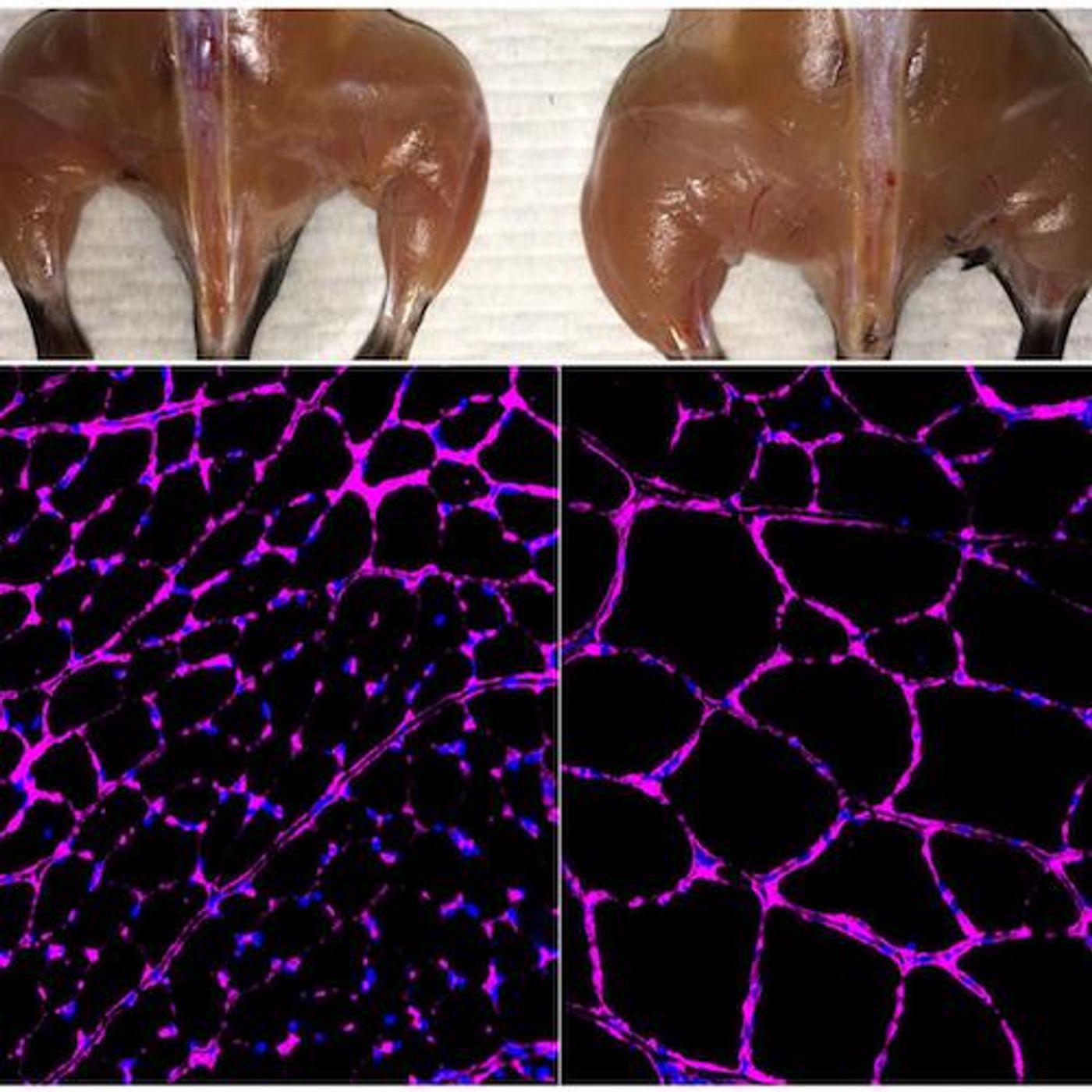
Incredibly, the scientists used their technique to reverse disease in several mouse models of disease. For acute kidney disease, the process activated genes that were previously damaged or silenced, and normal kidney function was restored. Liver cells were also induced to differentiate into cells that produce insulin in the pancreas, which partially rescued a mouse model of type 1 diabetes.
The researchers also recovered muscle growth and function in muscular dystrophy mouse models. Instead of trying to repair the disease-causing genetic mutation, they increased gene expression in the same cellular pathway that the defective gene works in, acting to over-ride the effect of the mutation. "We are not fixing the gene; the mutation is still there," explained Belmonte, "Instead, we are working on the epigenome and the mice recover the expression of other genes in the same pathway. That is enough to recover the muscle function of these mutant mice."
"We were very excited when we saw the results in mice," added co-first author Fumiyuki Hatanaka. "We can induce gene activation and at the same time see physiological changes."
They are continuing to refine the system, improving specificity and trying it in other kinds of cells. More work will be needed, but it could one day be part of human therapeutics.
Sources: Cell Press, AAAS/Eurekalert! Via Salk Institute, Cell