Depression affects millions of Americans and many of those people are taking some kind of medication. Selective serotonin reuptake inhibitors (SSRIs) are the most commonly prescribed antidepressant, but they are really not an ideal treatment. It can take 4-6 weeks for an SSRI to exert its beneficial effects, and for some living with depression, SSRIs don’t work at all. There are other treatments for depression (tricyclics, monoamine oxidase inhibitors, SNRIs), but they all act on the monoamine neurotransmitters (dopamine, serotonin, norepinephrine) and they can have horrible side effects. There is a lack of alternatives for treatment-resistant depression, and this is an especially big problem for those with comorbid alcoholism.
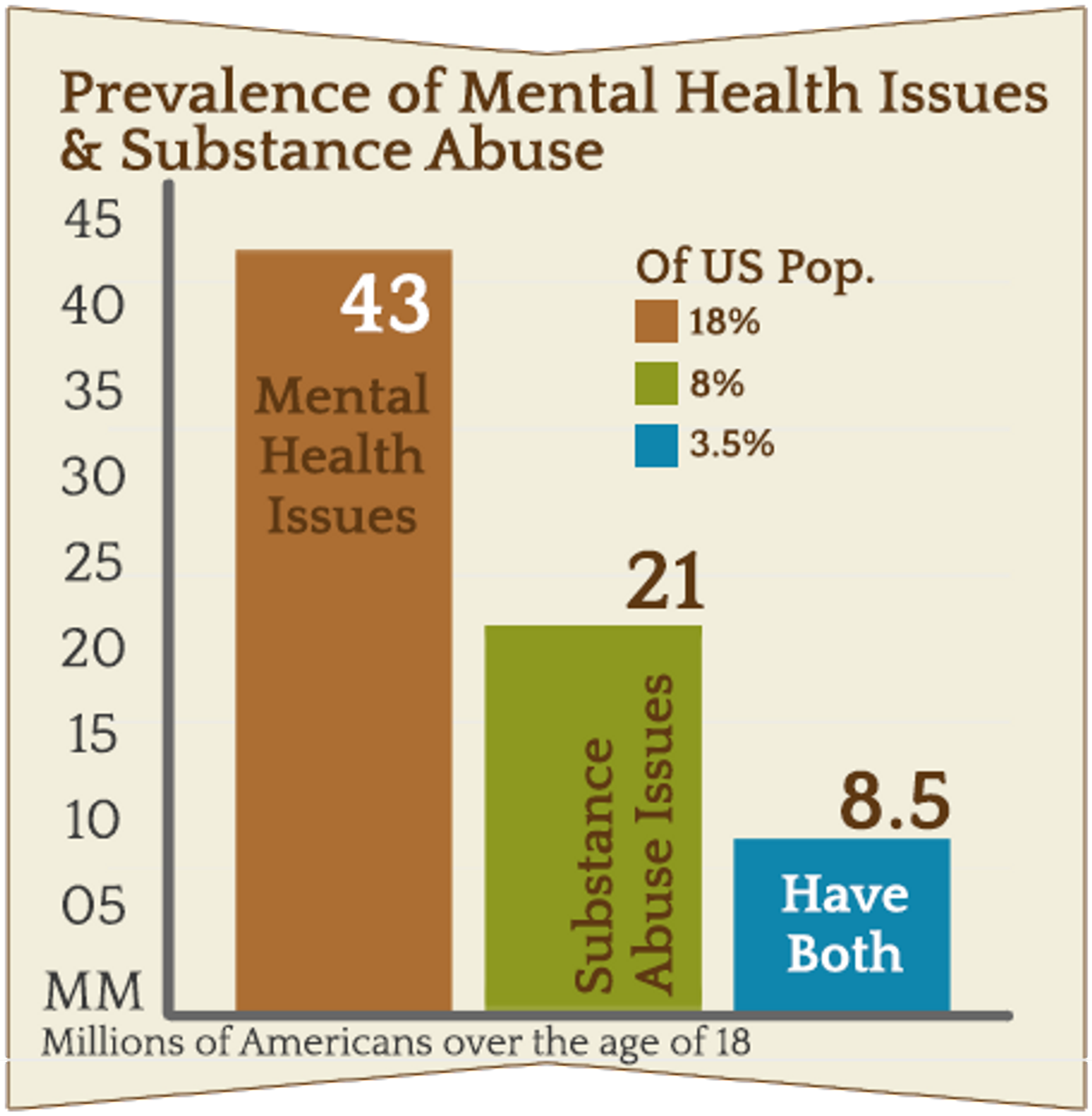
Alcoholism and depression are highly comorbid, but there is no united treatment for them. Usually, clinicians will treat one disorder and then the other, but that doesn’t account for the fact that the symptoms of one can make the symptoms of the other worse. Those with comorbid alcoholism and depression are more prone to be resistant to SSRI treatment, and so new therapeutics are needed to treat this especially vulnerable population.
Ketamine, a horse tranquilizer also known as the street drug “special k,” has been getting a lot of attention recently because it is a fast-acting antidepressant. Unlike current antidepressants that can take weeks to work, ketamine can alleviate symptoms of depression in hours. Researchers at
Vanderbilt University Medical Center have shown that, in an animal model of alcoholism-induced depression, ketamine administration can ameliorate symptoms of depression. Ketamine is a NMDA receptor antagonist, but it is competitive with alcohol at this receptor. What that means is, in addition to its “dissociative anesthetic” effects, ketamine has high abuse potential for alcoholics.
Because of this significant downside of ketamine, the Vanderbilt group also tested a monoacylglycerol lipase (MAG) inhibitor. MAG lipase degrades 2-AG (2-arachidonoylglycerol), one of the endogenous ligands for the CB1 receptor. The endocannabinoid system has been implicated in depression and anxiety and, specifically, the Vanderbilt researchers showed that following alcohol exposure, there are elevated levels of 2-AG in parts of the brain that are involved in those disorders. Pharmacologically raising the levels of 2-AG to activate the CB1 receptor could alleviate the depressive symptoms and possibly reduce the cravings that come with withdrawal. Using the same animal model of alcohol-induced depression, treatment with a MAG lipase inhibitor reduced depressive behavior.
An upside of using an enzyme inhibitor to increase the amount of an endogenous agonist is that there will be no psychotropic effects. THC, the active compound in marijuana, is also a CB1 agonist, but the psychotropic effects aren’t desirable in a clinical setting. Another benefit of targeting CB1 is that alcohol does not bind to or affect this receptor at all. Unlike ketamine which can substitute for alcohol, cannabinoids, endogenous or otherwise, cannot, meaning that there is much, much lower abuse potential for alcoholics.
It is exciting to see researchers finally moving beyond the monoamine-focused treatments for depression. SSRIs and other similar drugs have helped so many people, but there are certain populations that these drugs don’t help. Treating comorbid alcoholism and depression or substance use disorder and depression is very difficult and requires a more integrated treatment plan. This a vulnerable population who would greatly benefit from a more effective therapeutic.









