Researchers have transformed mouse fibroblasts, the most common type of cell in mammalian connective tissue, into induced cardiac progenitor cells.
Researchers at the University of Wisconsin found there were 11 genes pivotal in embryonic heart development that could be used to genetically reprogram fibroblasts. The team narrowed down the number of necessary genes from 11 to five.
They found the necessary conditions for the cells to be effectively transformed and cultured in a lab setting. Then, from the five genes, they developmentally rewound the fibroblasts cells back in time and transformed the common cells into cardiac muscle cells, smooth muscle cells, and endothelial cells.
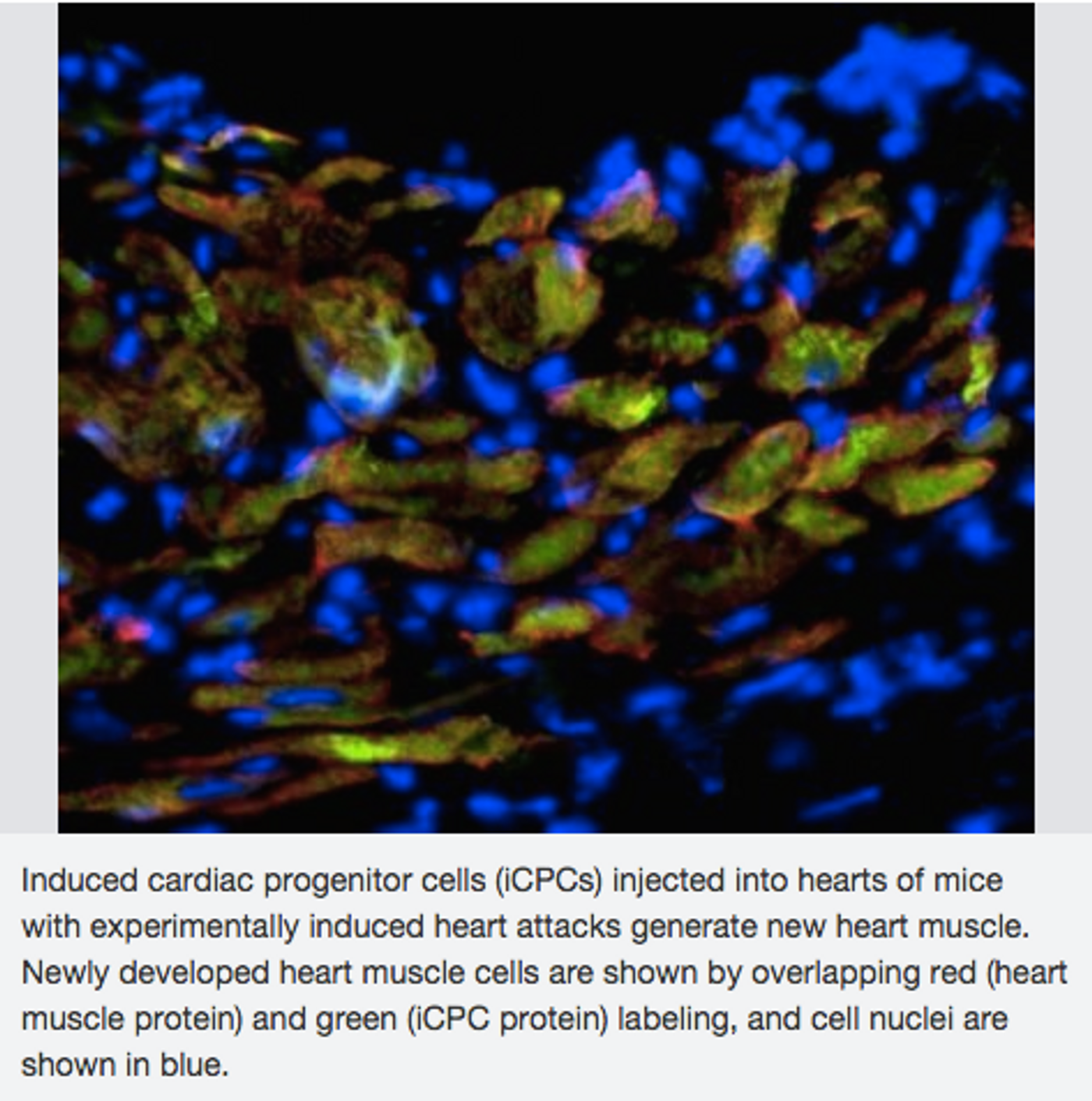
The researchers then tested the new cells on mice with induced heart attacks. They injected the cells into the damaged mice hearts. The engineered cells moved towards the damaged areas of the heart and created the three critical heart cell types, thus generating new heart muscle. The mice with the injected heart cells were more likely to survive than the mice without the injected cells.
Like stem cells, progenitor cells have the ability to replicate. However, in comparison to stem cells, “you can reduce the risk of tumor formation,” said cardiologist Timothy J. Kamp, who led the research team. Stem cells can turn into any of the 220 types of human cells. However, “[cardiac progenitor cells] are more committed to the heart lineages and are unlikely to form a tumor,” Kamp said.
The discovery could allow the development of a scalable method of creating a nearly unlimited supply of the three major types of heart cells. The billions of cells would be enough material to study heart disease in laboratory dishes. If the finding can be replicated in human cells, the discovery could jumpstart the development of new drugs, novel models for heart disease, and raw materials for treating diseased hearts by replacing diseased cells with healthy cells.
The research was published on February 11, 2016, in the journal
Cell Stem Cell.
 Researchers have transformed mouse fibroblasts, the most common type of cell in mammalian connective tissue, into induced cardiac progenitor cells.
Researchers have transformed mouse fibroblasts, the most common type of cell in mammalian connective tissue, into induced cardiac progenitor cells.