How does estrogen increase the risk of autoimmunity for women?
The unique activity of estrogen hormones and their cell receptors in the female body may explain why women are more likely than men to develop autoimmune diseases. From the University of Turku, scientists investigate an estrogen pathway as a new way to target autoimmunity.
In general, women are more likely than men to develop autoimmune diseases, like rheumatoid arthritis, multiple sclerosis, and lupus – making estrogen a likely causative factor in autoimmunity. In the female body, estrogen is the key hormone for growth and development of female sexual characteristics and reproduction.
The name “estrogen” includes several related hormones, such as estrone, estradiol, and estriol. Estrogen is produced in the ovaries, adrenal glands, and fat tissues, circulating in the blood and binding to cells via hormone receptors on the cell surface. After binding, estrogen activity has a variety of effects on breast and uterus tissue as well as brain, bone, liver, and heart tissue. For example, estrogen is responsible for regulating the growth of the uterine lining in the first stage of the menstrual cycle.
However, estrogen is not completely unique to females. In males, estrogen plays a role in modulating libido, erectile function, and spermatogenesis.
Estrogen binds cells through the estrogen receptor alpha protein. This is one estrogen receptor subtype with activity in the nucleus. Mainly, this receptor helps transcribe certain target genes, giving it the ability to regulate key physiological functions in the mammary glands, uterus, ovaries, bones, liver, and adipose tissue. This receptor’s “cousin,” estrogen receptor beta protein, is found in bladder, ovary, colon, and adipose tissue.
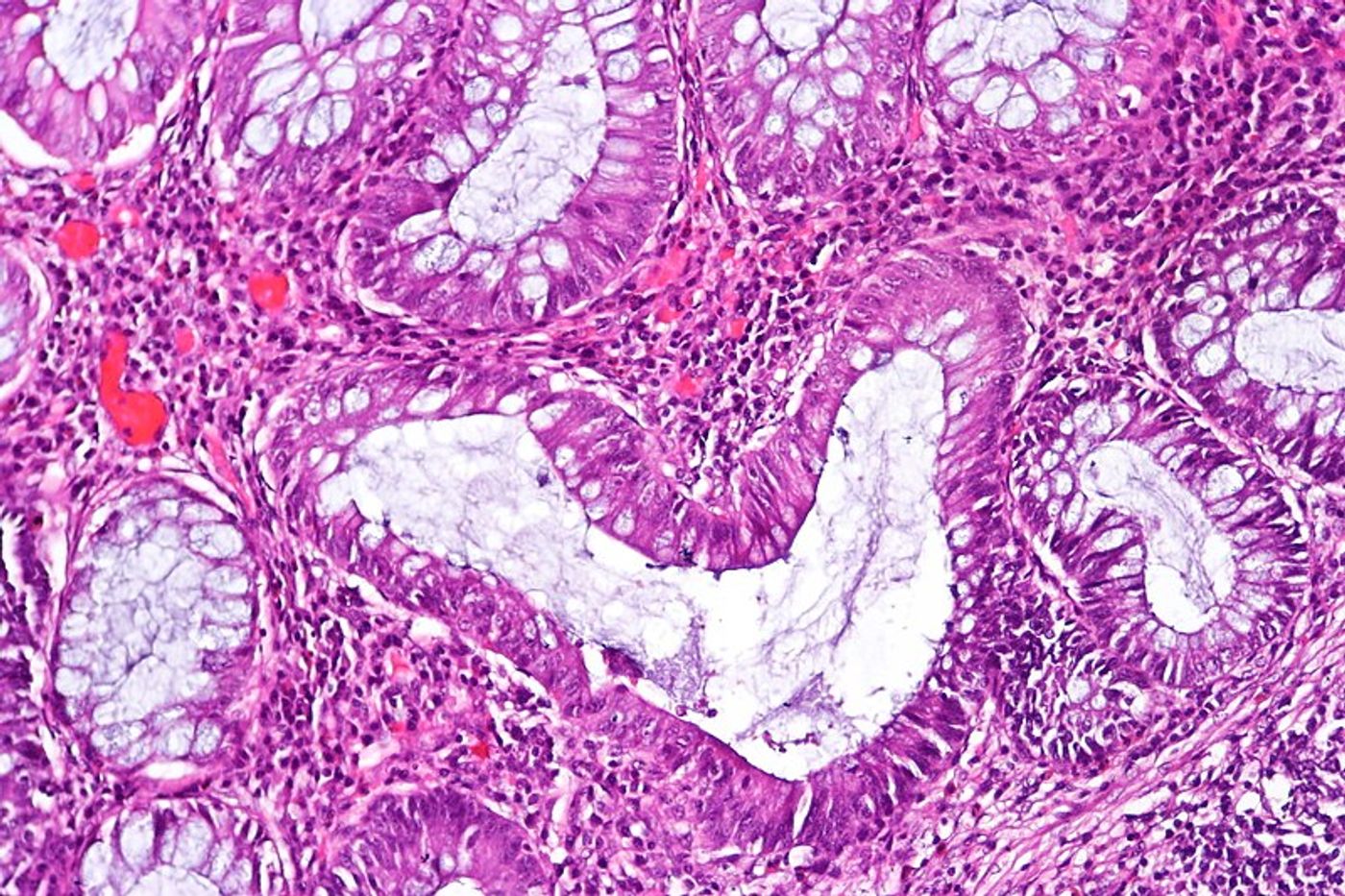
So what happens when T cells’ estrogen receptors are missing? With their new study, scientists wanted to answer this question. Thus, they genetically altered mice to lack estrogen receptor alpha protein on T cells. When they transferred these lacking T cells to mouse models of human inflammatory bowel disease, the mice did not develop inflammation in the colon, compared to mouse models without the transferred cells who did develop disease.
RNA sequencing studies also helped them find that the estrogen receptor alpha protein “regulates multiple aspects of T cell function, including T cell activation, proliferation, and survival,” explained research leader Docent Zhi Chen.
With this finding, it is likely that the receptor protein is causing dysfunction in regulatory T cell activity. These “Tregs” are vital for maintaining the balance in the immune system. Too little immune activity leads to pathogenic invasions running rampant in the body, and too much immune activity results in allergies and autoimmune diseases. It could be that estrogen receptors are disrupting that balance, explaining in part why women are more vulnerable to autoimmune diseases than men.
The present study was published in the journal Science Signaling.
Sources: HealthyWomen.org, Steroids, Asian Journal of Andrology, University of Turku