Revealing How a Common Virus Evades the Immune System
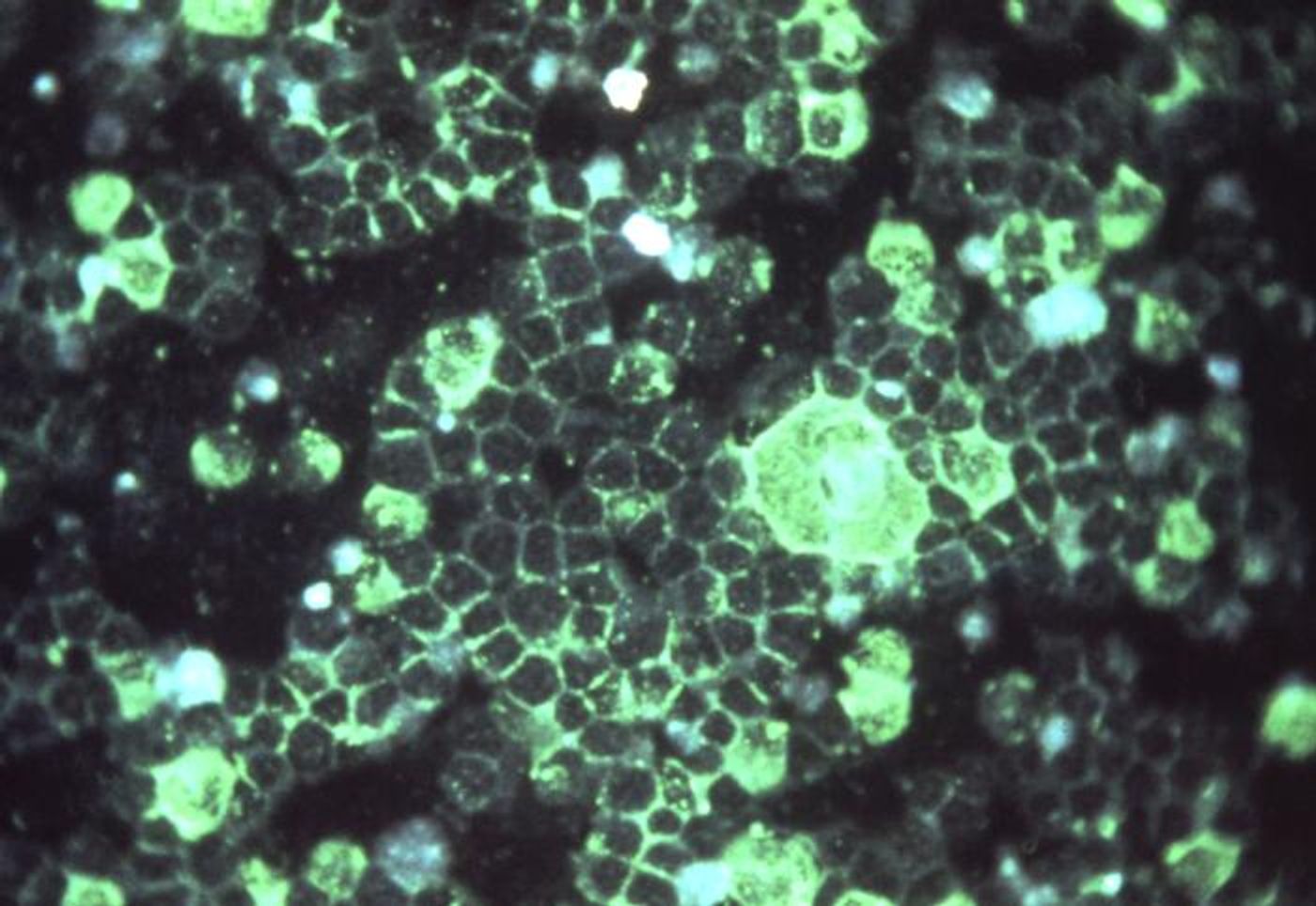
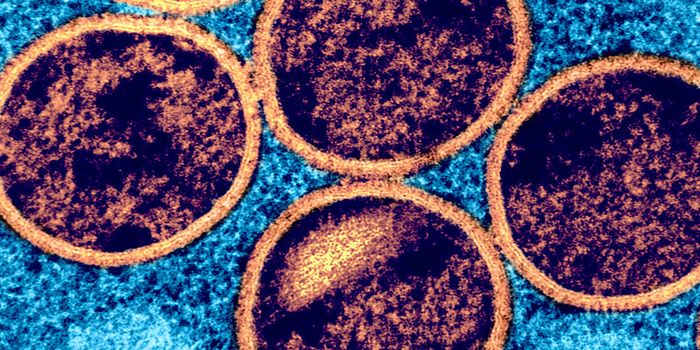
The human metapneumovirus (HMPV) might not be well-known, but it is the second biggest cause of respiratory infections, which can be especially serious for infants and the elderly. This virus is related to respiratory syncytial virus (RSV), the leading cause of respiratory infections. Scientists have now learned how HMPV remains undetected by the immune system. This work, which was reported in Nature Microbiology, may aid in the development of a vaccine for both HPMV and RSV.
"This is exciting because RSV was discovered in 1953, but we still don't have a vaccine. The virus inhibits the innate immune response, and can infect the same person again and again," said the senior study author Jianrong Li, a professor of virology in The Ohio State University Department of Veterinary Biosciences. "Now we have a mutant strain of HMPV that can trigger a higher immune response. Right now we're working to translate this same concept to see if it can work for RSV, too."
The genomic information of many viruses is encoded not in DNA, but in RNA. HMPV is an RNA virus, and the scientists found that it takes advantage of an RNA modification to hide from the body's first line of defense against pathogens, called the innate immune response. Thus, the virus can replicate and cause an infection. Using a rat model, the researchers found that a mutated version of the virus works as a vaccine against HPMV; it triggered an innate and adaptive immune response.
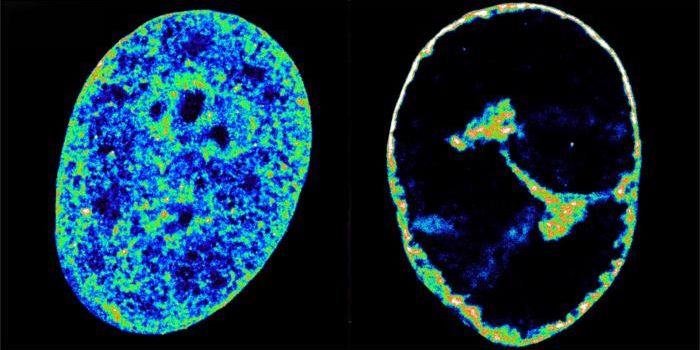
The Li lab wanted to know whether HPMV carries a common type of epigenetic modification that is seen on RNA, called an N6-methyladenosine (m6A) modification. They used a technique called high-throughput sequencing to find the viral gene that carried the most m6A methylation and then removed them to see the impact. The mutated virus stimulated the production of an antiviral molecule called type I interferon, showing that the innate immune response was initiated, and in a more robust way compared to the unmutated virus.
"This opened up a big question," Li said. "Why would a virus lacking this methylation produce a much higher innate immune response?"
The m6A modification shields the virus from the immune system by disrupting the ability of immune proteins to tell the difference between RNA from a virus, known as nonself-RNA, and RNA made by host cells, called self-RNA.
"We know that when a virus infects cells, it produces RNA, and human cells in the innate response try to separate self-RNA and nonself-RNA," Li said. "The virus is smart: It gained this methylation, and now our host innate response is confused. That's how the virus escapes recognition by the innate immune response."
"We are very excited about this finding. This novel function of m6A may also be conserved in many viruses," said the first study author Mijia Lu, a postdoctoral researcher in Li's laboratory.
"In the case of cotton rats, the mutant virus produced a higher amount of type I interferon and triggered a higher antibody response and a higher T-cell immune response. That means you've triggered higher protective ability against the virus infection. So mutating the virus enhances vaccine efficacy," Li said. "That is exactly what we want. We proved a concept that these mutant viruses are improved vaccine candidates for HMPV."
Sources: Phys.org via The Ohio State University, Nature Microbiology