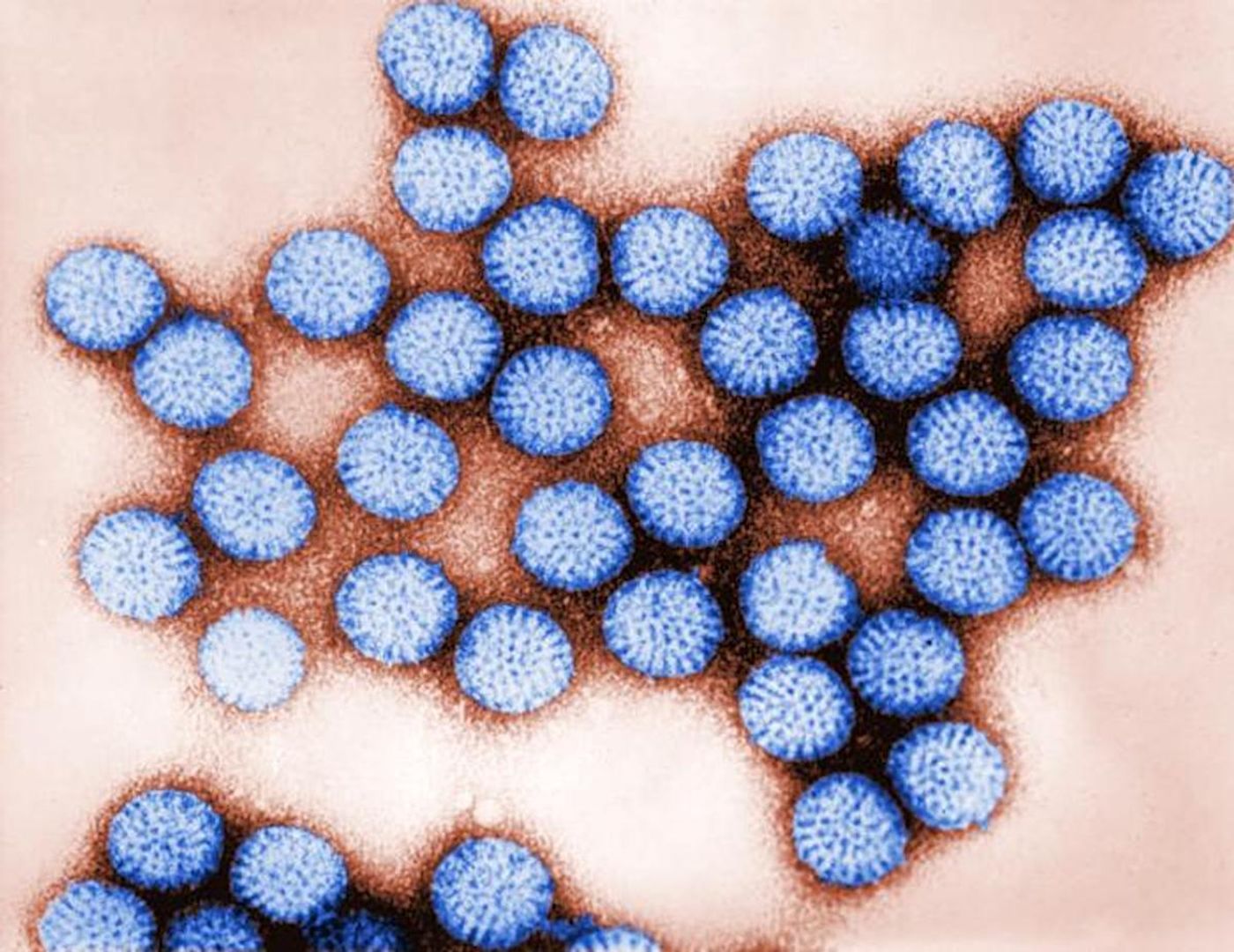
The Mechanisms Underlying Rotavirus Infections are Revealed
According to the World Health Organization, globally, rotaviruses are the biggest reason why young children get diarrhea. It's estimated that 215,000 children younger than five die every year from rotavirus infections, which are now preventable with one of four vaccines.
Rotaviruses infect cells in the small intestine called enterocytes, and the virus can cause disease in other cells that aren't infected with it. It's been suggested that infected cells are releasing signals that are interfering with the function of uninfected cells.
Reporting in Science, researchers have now found that these rotavirus-infected cells release adenosine diphosphate (ADP), causing the activation of a molecule called P2Y1, which triggers intercellular calcium waves in uninfected cells.
The scientists used a mouse model of rotavirus infection to show that diarrhea severity could be reduced by disrupting the binding between ADP and P2Y1. This may open a treatment avenue for infected people.
"In our previous studies using fluorescent calcium sensors and time-lapse imaging, we discovered that rotavirus-infected cells display aberrant calcium signals that we can visualize as bright pulses of intercellular calcium waves that radiate from the infected cells," said the corresponding study author Dr. Joseph Hyser, assistant professor of virology and microbiology and member of the Alkek Center for Metagenomic and Microbiome Research at Baylor College of Medicine. "Calcium signaling was known to be associated with various aspects of rotavirus infection and our work revealed the dynamic nature of the alterations induced by rotavirus."
This work utilized several models including monkey and human cell lines. The researchers tested the ability of various molecules to cause calcium waves in the cells. Some candidate molecules, like prostaglandin E2 and nitric oxide, didn't affect calcium. However, ADP and ATP, which are known to play roles in calcium signaling but have not been linked to rotavirus infection, were implicated. The researchers deleted the expression of the P2Y1 gene, which prevented ADP signaling and lowered intracellular calcium waves. Rotavirus-infected cells were found to initiate these waves by releasing ADP, which binds to P2Y1 receptors on neighboring cells that are not infected.
"Across the three model systems we consistently found evidence that rotavirus-infected cells signal uninfected cells with ADP and that this contributes to the severity of the disease," said first study author Dr. Alexandra L. Chang-Graham, who is completing an M.D./Ph.D. program. "We consider it paradigm-shifting that the actual signal, ADP, was not even on the radar before."
Additional work revealed that ADP signaling also raises the severity of infection by increasing the expression of cytokine IL1-alpha, promoting inflammation, and increasing the secretion of serotonin, worsening diarrhea. When ADP signaling was disrupted in cells, intercellular calcium waves were reduced, as were the release of the compounds.
"Finally, we determined that inhibiting the P2Y1 receptor reduced the severity of rotavirus-induced diarrhea in a mouse model," Chang-Graham said. "Using intercellular calcium waves, rotavirus amplifies its ability to cause disease beyond the cells it directly infects. This is the first virus identified to activate ADP-mediated intercellular calcium waves. This may be a strategy that other viruses also use to cause disease in their hosts."
"Our findings add a new and very potent signaling pathway into the causative mechanisms of rotavirus diarrhea," Hyser said. "In terms of treatment, this is exciting because currently, some drugs targeting P2Y1 are undergoing preclinical testing as anticlotting drugs. It's possible that such drugs could be repurposed if proven to be safe for children, to be used to treat diarrhea caused by rotavirus infection."
Sources: AAAS/Eurekalert! via Baylor College of Medicine, Science