Immune Cells Use Bleach to Destroy Bacteria
Scientists at Ruhr-Universität Bochum have learned more about how immune cells use a toxic cocktail to battle bacteria. They determined that incredibly, chlorine bleach is one of the ingredients in the mixture. The proteins in affected bacterial cells get doused with reactive oxygen, causing the destruction of the bacterium. The findings have been reported in eLife.
Cells of the immune system spring into action when bacteria enters our bloodstream. The most common kind of white blood cell, neutrophil granulocytes, are the first line of defense and can devour the microbes. After exposure to a toxic brew of chemicals including hydrogen peroxide, antiseptics, and chlorine bleach, oxidation alters the bacteria, killing it. While we know the basics of the process, many details are still unknown, leaving questions unanswered.
"Once we'll find an answer to these questions, we will understand how some bacteria outwit our immune system and how the immune system is impaired by certain genetic defects," explained researcher Lars Leichert, head of the research group Microbial Biochemistry.
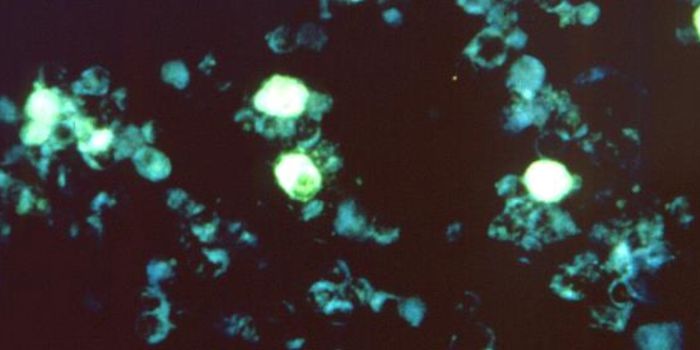
For this work, the researchers utilized recently created fluorescent proteins. They carry a signal that indicates chemical oxidation states. When exposed to blue light, these proteins, called roGFPs, glow green. If oxidized, they also glow under violet light.
The scientists used Escherichia coli bacteria that were engineered to carry genes encoding for roGFPs. After introducing these bacteria to immune cells, the bacteria was engulfed as the researchers expected. With a super-resolution microscope, the investigators saw that the roGFPs within the bacteria glowed after being exposed to blue light. After they had been completely devoured, it took only seconds for the oxidation of the roGFPs, which then glowed under violet light.
"Based on the speed and the specificity with which the roGFPs oxidized, we have deduced that chlorine bleach plays the key role in the process. That means immune cells utilize bleach to kill bacteria," concluded Leichert.
To find enzymes important to the mixing of the deadly cocktail, the scientists created cells in which certain enzymes were missing. It was found that when the enzyme NOX2 was absent, such altered cells couldn’t kill bacteria by oxidation. Myeloperoxidase is an enzyme necessary to generate chlorine bleach, and when it was blocked, there was also a marked reaction in oxidation.
Check out the following video from the research.
Sources: AAAS/Eurekalert via Ruhr-Universität Bochum, eLife