A New Type of Antibiotic has Been Found
Now that people are facing a crisis from the rise of antibiotic-resistant bacteria, there has been considerable research interest in identifying new drug candidates to help stem the problem (learn more about the issue from the video). Now, an international team of researchers has reported the discovery of a new class of antibiotics, called odilorhabins or ODLs, which are made by bacteria that live within nematode worms. These new molecules have a unique way of killing bacteria, which may also be useful for eliminating bacteria that carry drug resistance genes. The work, reported in Molecular Cell by investigators at the University of Illinois at Chicago and Nosopharm, a Lyon, France biotechnology company, outlines the antibiotic as well as its mode of action.
These symbiotic bacteria that produce the antibiotic are not well understood. The Nosopharm scientists assessed 80 strains of it to look for antimicrobial characteristics. Compounds that displayed activity were then isolated, and the researchers could study the structure of those compounds. Derivatives that were even more powerful could then be created.
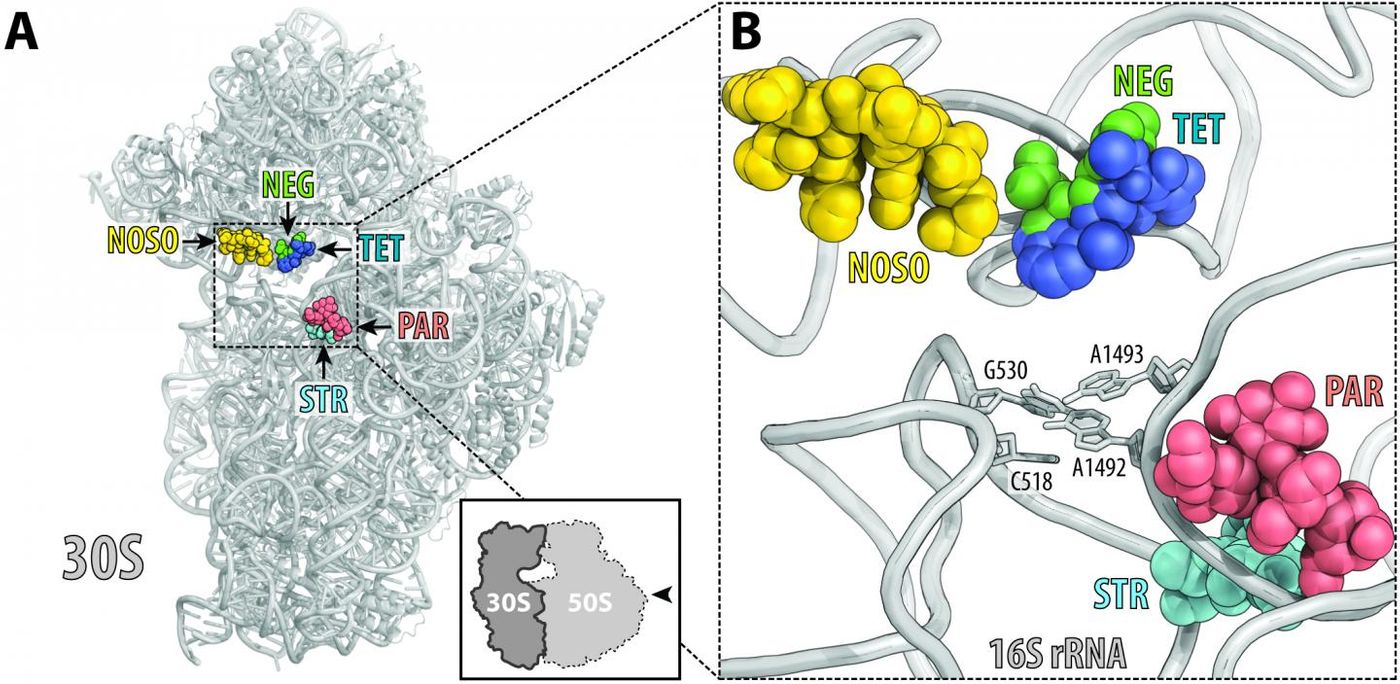
Corresponding authors Alexander Mankin and Yury Polikanov of UIC led the study on the drug's mechanism of action. ODLs exert their effect on an organelle called the ribosome, which builds proteins for the cell, and in this case, the bacterium.
"Like many clinically useful antibiotics, ODLs work by targeting the ribosome, but ODLs are unique because they bind to a place on the ribosome that has never been used by other known antibiotics” explained Polikanov, assistant professor of biological sciences in the UIC College of Liberal Arts and Sciences.
"When ODLs are introduced to the bacterial cells, they impact the reading ability of the ribosome and cause the ribosome to make mistakes when it creates new proteins," said Mankin, director of the Center for Biomolecular Sciences in the UIC College of Pharmacy. "This miscoding corrupts the cell with flawed proteins and causes the bacterial cell to die."
Many antibiotics act to slow the growth of bacteria, and Mankin noted that antibiotics that kill bacteria directly, bactericidal antibiotics, are unusual.
"The bactericidal mechanism of ODLs and the fact that they bind to a site on the ribosome not exploited by any known antibiotic are very strong indicators that ODLs have the potential to treat infections that are unresponsive to other antibiotics," said Mankin.
The Nosopharm team assessed how well the ODL compounds acted on bacterial pathogens, including strains that are known to develop resistance.
"We found that the ODL compounds cured mice infected with several pathogenic bacteria and demonstrated activity against both Gram-negative and Gram-positive pathogens, notably including carbapenem-resistant Enterobacteriaceae," revealed co-corresponding author Maxime Gualtieri, co-founder and chief scientific officer of Nosopharm. (Learn more about these deadly bacteria from the following video.)
The researchers think that this work highlights the value of international collaboration when confronted with serious problems. "In this case, the combined expertise of the industry, which has the resources to develop new compounds, and the academia, which has the research and lab capabilities to understand how the compounds work, have enabled the development of this new class of antibiotics," Mankin noted.
"The collaboration between multiple labs with diverse areas of expertise add credibility to the research," Polikanov added.
A lot of work remains; the therapeutic potential and possible drawbacks of ODL have to be assessed thoroughly, but these findings show that such a study is warranted.
"Collaborations with academia with renown expertise in antibiotics like the UIC team help us for this preclinical development," said Philippe Villain-Guillot, co-founder and chief executive officer of Nosopharm.
Sources: AAAS/Eurekalert! Via University of Illinois, Molecular Cell