Key Markers in Melanoma Disease Progression
We know that we’re supposed to watch our existing moles for change throughout our lives. We know to look for what is normal and to watch for changes that look “weird”, “angry”, “black or multi-colored”, or for “strange shapes/borders”. But what happens before and while those changes take place? When does a mole transition from being just a mole to something cancerous like melanoma?
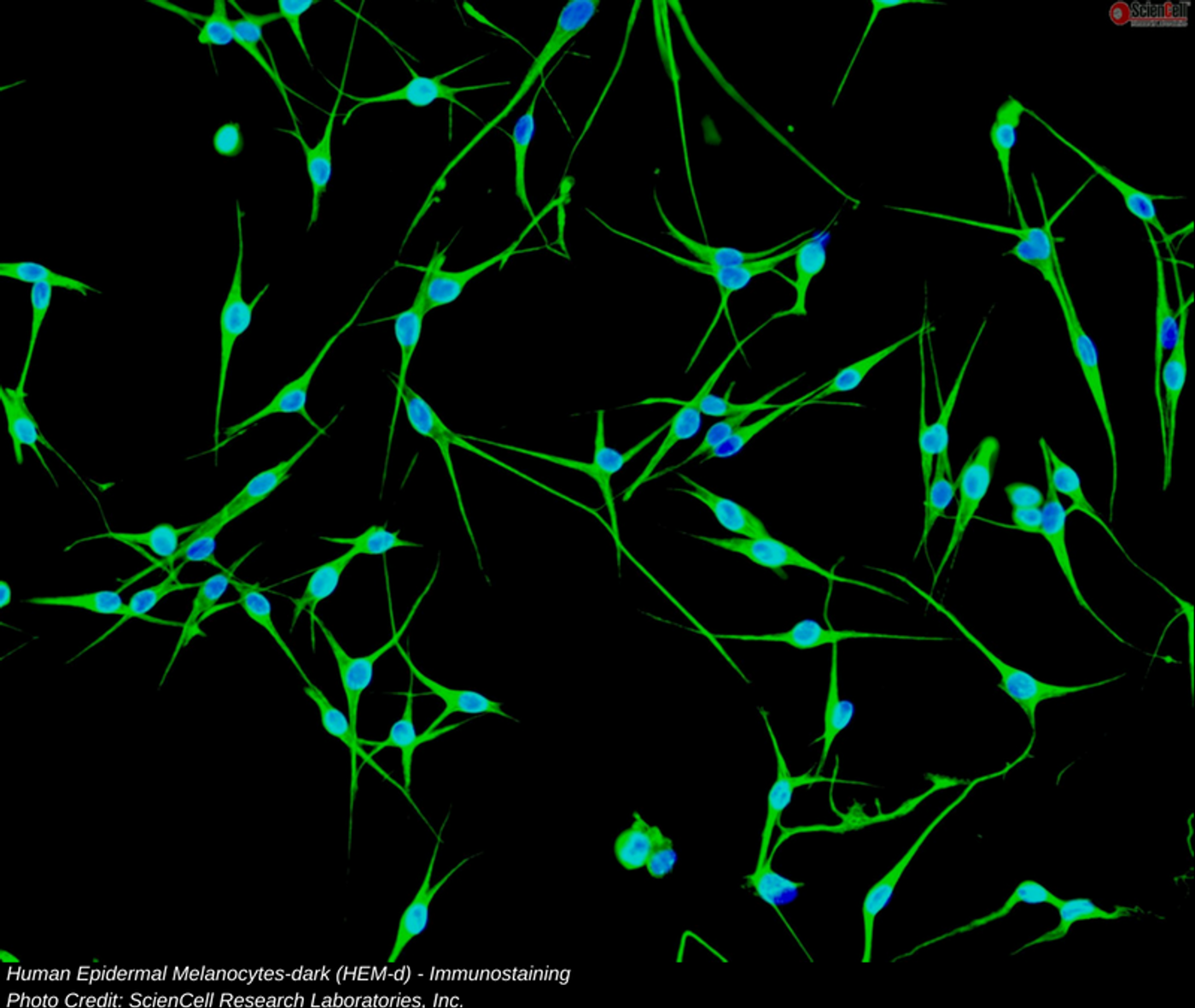
Researchers at the University of California San Francisco utilized CRISPR to reconstruct the process of melanoma growth from one skin cell to the next in a laboratory environment. Their goal was to identify any critical molecular indications that could be extrapolated and ultimately found in vivo as early warnings of cancerous change in humans. The group also hoped to find targets for future therapies that may help with early cancer progression and provide better outcomes for melanoma patients. Skin cancer is the most common type of cancer diagnosed in the world; melanoma is less common (5% of skin cancers) than some other types of skin cancer but has the higher likelihood of growing, spreading, and causing death. Melanoma can spread very quickly.
Melanoma has a distinct development pattern characterized in the literature by the following stages: benign nevus, dysplastic nevus, radial-growth phase, vertical-growth phase, to metastatic melanoma which dissociates form the primary tumor and then goes on to grow at distant sites. Researchers looked at independent gene mutations involved in molecular signaling pathways regulating cell growth, replication, proliferation, and progression to further disease. The group suspects that rather than a “light switch” type of change that creates malignant growth, the progression is subtle, in molecular terms, with an additive type of effect that does not necessarily set off our body’s immune system alarms; this allows slower ramping up of genetic changes so that at the time the cell’s individual danger signals should go off, they are overwhelmed and may not even function correctly.
In the first part of their research, the researchers found that somatic alterations turned on the mitogen-activated protein kinase (MAPK) signaling pathway, induced increases in telomerase production, developed an override functionality for the G1/S checkpoint in the replication cycle, and interrupted the p53 signal cascade. The group determined that point mutations caused by UV-radiation continued until melanoma invasion occurs.
CDKN2A tumor suppressor loss is linked to metastatic melanoma, as previously studied in the literature, however the process and mechanisms leading to this loss are not widely understood. In the second part of their research, the group used CRISPR-Cas9 to identify a transcription factor downstream of CDKN2A, called BRN2; this factor’s increased expression coincides with the CDKN2A loss which is also the final break in regulation that acts to inhibit invasive expansion. The group concluded that CDKN2A inhibits BRN2 and this structure keeps melanoma from becoming invasive and metastasizing.
The hope is that this information can better identify patients in a timelier manner for prognosis and treatment planning. Additionally, these findings may be an opportunity for testing to be developed to assess a patient’s risk of skin cancer beyond the checking of moles externally.
Sources: UCSF, Cancer Cell-part 1, Cancer Cell-part 2 , American Cancer Society,