Why Automation is Outpacing Traditional Methods for Single-Cell Cloning
Genetic modification of mammalian cells has vast use in biotherapeutics development and basic research applications. An important step early on is the isolation of clones with the desired mutation prior to downstream analysis. While fluorescence-activated cell sorting (FACS) and limiting dilution have traditionally been the methods of choice, many are turning to automated, image-based solutions.
The importance of monoclonality
Whether you’re studying the effects of a gene knockout in a signalling pathway or developing a therapeutic antibody, you start by introducing a mutation into your cell line of choice. There are various advanced gene editing technologies to choose from, such as CRISPR/Cas9, which has made it possible to alter genomes with relative ease.
But no method is perfect. Successful targeting of cells depends on transfection efficiency, cell type, and other factors. That’s why you typically end up with some proportion of cells that did not acquire the desired mutation. Single-cell isolation techniques are used to separate individual cells with the desired mutation from a heterogeneous background before further experimentation.
What is limiting dilution?
Limiting dilution is a basic technique for generating a monoclonal cell line from a mixed cell population. It works based on the principal of Poisson distribution; essentially cells are serially diluted until you have a high probability of plating a volume that contains only one cell. Sometimes, multiple rounds are needed to be sure you actually have one clone.
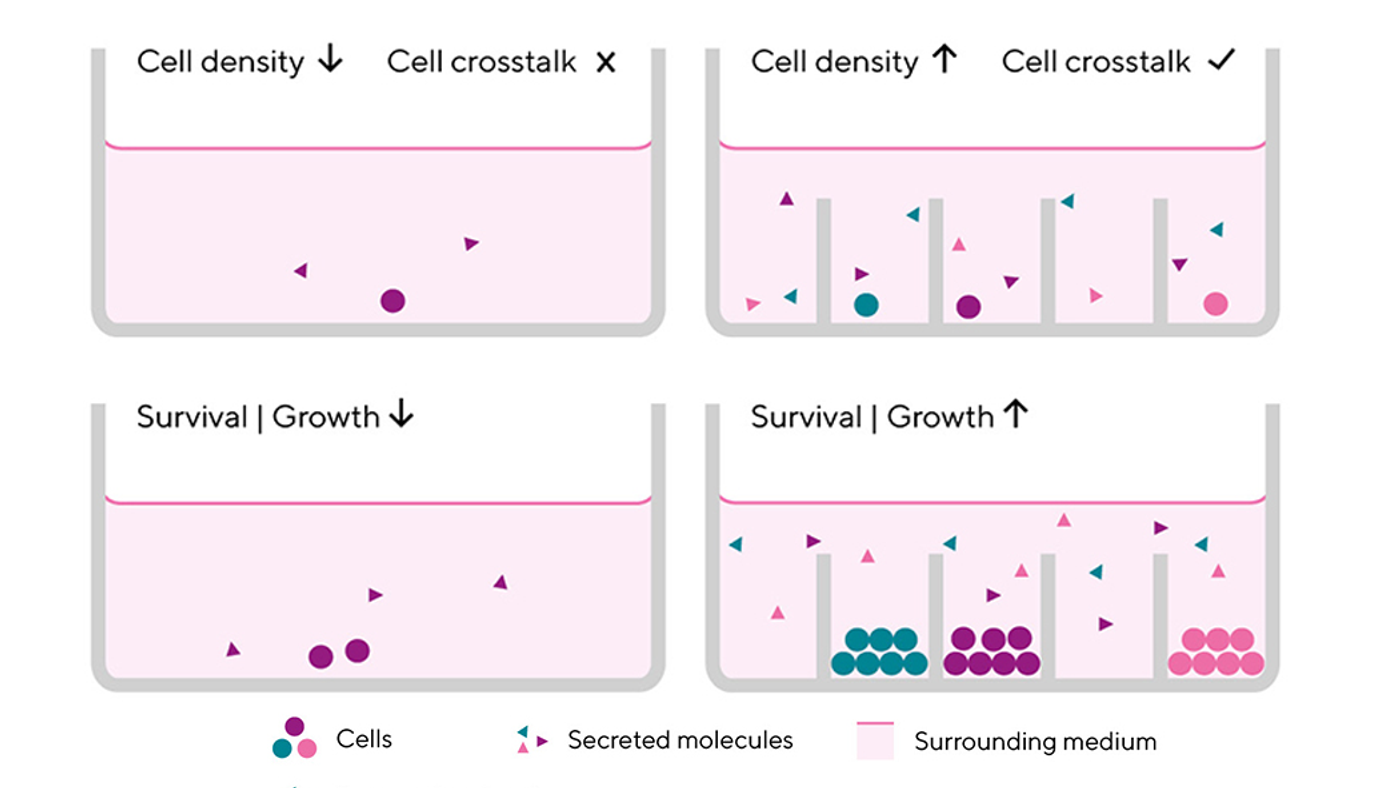
It’s well known that single cell cloning by limiting dilution is time consuming and difficult, but scientists choose it because it does not involve complicated lab equipment. Still, the process takes weeks to months and lots of manual steps without any guarantees. Limiting dilution is also stressful to cells because it deprives cells of crosstalk via growth-promoting factors in the media.
In industrial applications where speed and monoclonality matter, limiting dilution can be limiting.
Cell lines used for the development and commercial production of biotherapeutics face additional scrutiny to ensure stability, monoclonality, and product quality. If not caught early, genomic heterogeneity in the cell line can lead to costly setbacks later on.
As part of the regulatory compliance process, cell line development workflows must provide proof of monoclonality, often requiring visual documentation. This is difficult to achieve with traditional techniques for single cell isolation like FACS and limiting dilution.
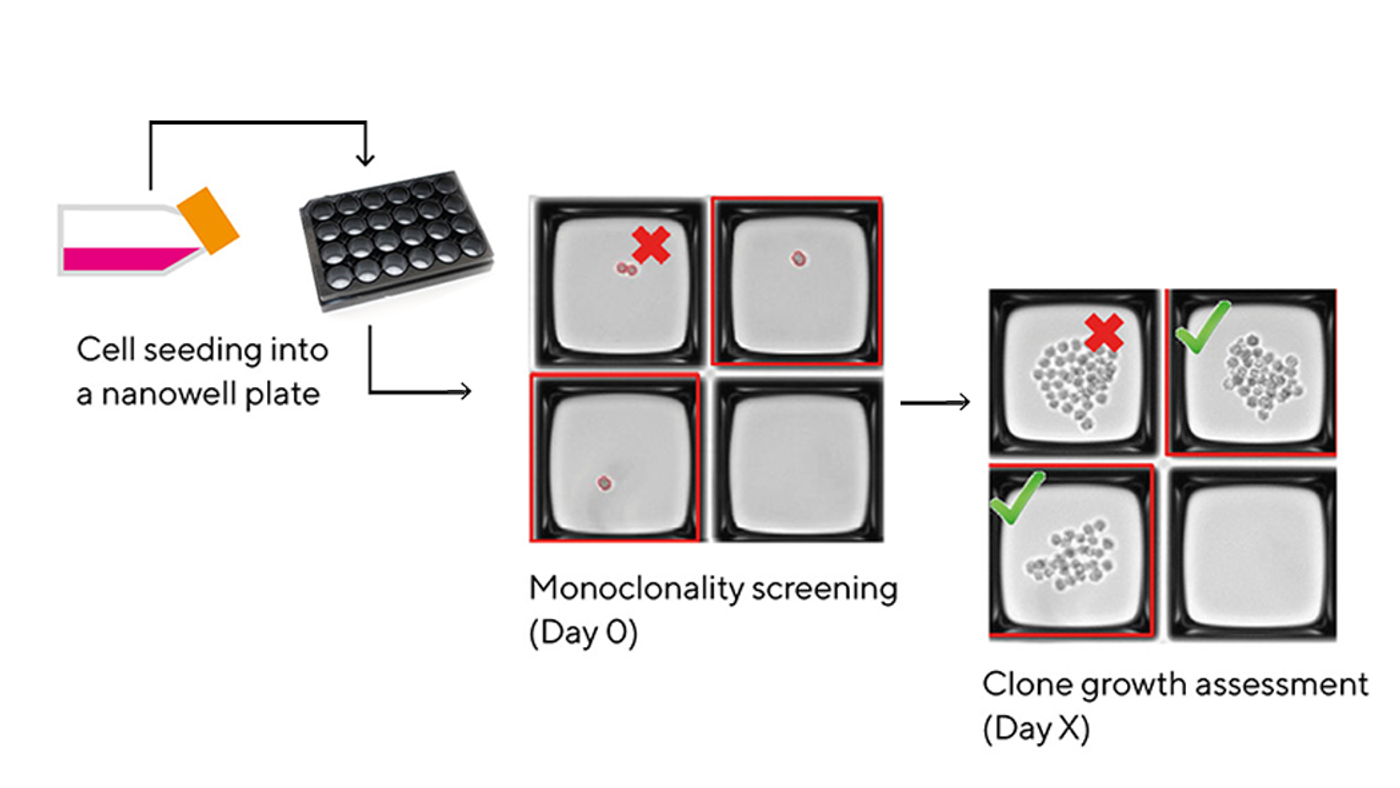
New image-based tools for single cell isolation and cloning fully automate the process, while also providing full compliance support where needed. With the CellCelector Flex platform, for example, you can get monoclonal, viable, and productive colonies from your pool of single cells in less than a week.
Not only can advanced systems for single cell isolation save you consumable, media costs, and incubator space, but they can also help avoid missteps and unnecessary procedures. With the CellCelector Flex, you also get extra assurances thanks to complete workflow documentation and high-quality in-process image-verified monoclonality proof.
View this Application Guide to learn about the benefits of the CellCelector Flex platform for single-cell isolation and picking in a range of single-cell applications including circulating tumor cells, fetal cell isolation and CRISPR single-cell cloning.