S1P1 Gene Mutation to Blame for Lethal Heart Condition
While studying the formation of the kidney, researchers realized that a gene they had deleted to examine the effect of its absence on kidney function also had negative impacts elsewhere in the body: the heart.
"The heart is the first organ that develops, and so when we deleted this gene in these precursor cells, we found that it resulted in abnormalities of the heart, severe edema, hemorrhage and low heart rate,” explained lead researcher Maria Luisa S. Sequeira-Lopez, MD.
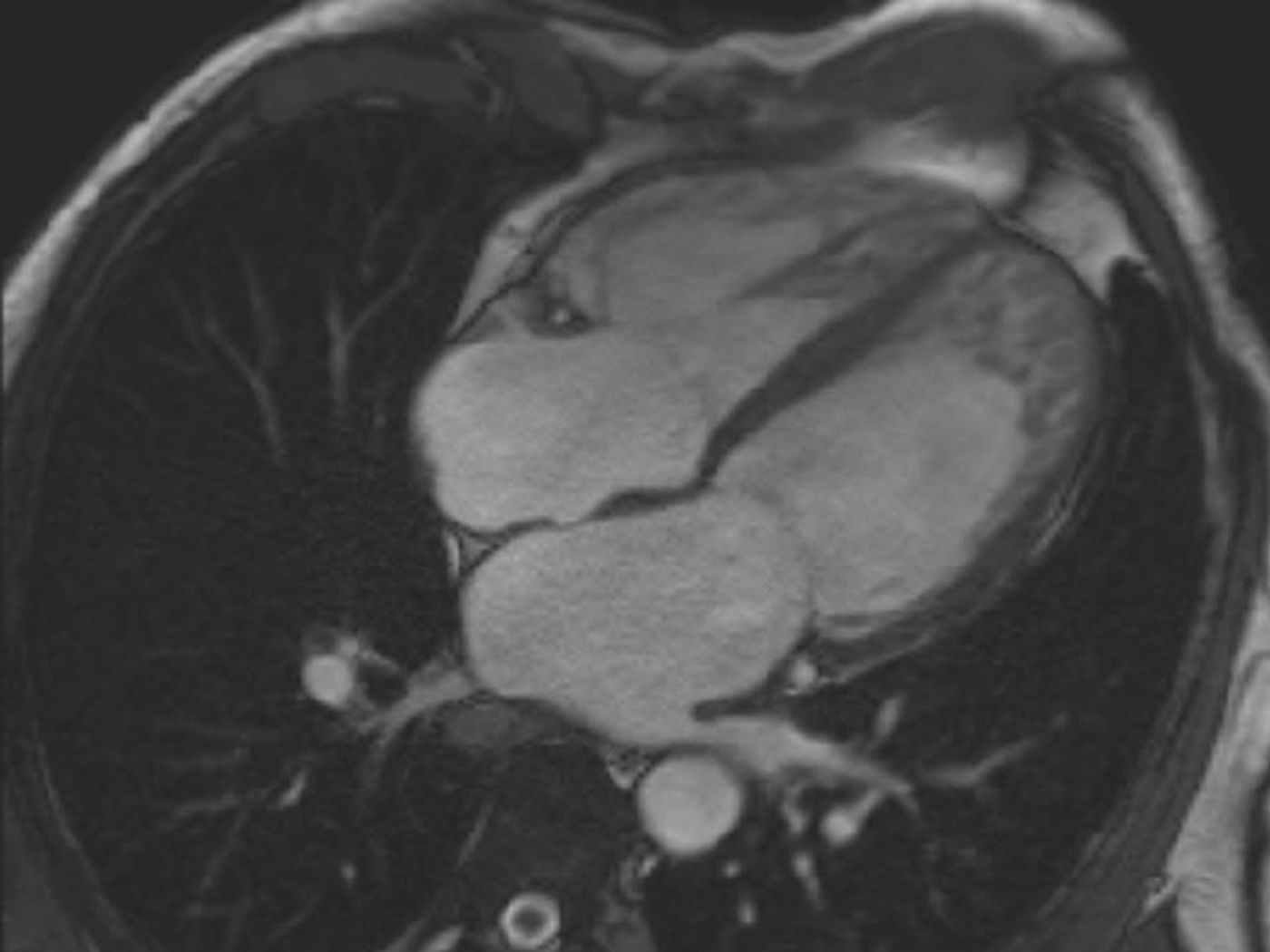
The situation Sequeira-Lopez described is a the beginning of a condition called ventricular non-compaction cardiomyopathy. The gene, called S1P1, is required for the production of heart tissue that adequately facilitates contraction and blood pumping. Without functional S1P1, a person is often prone to early death. “There is an urgent need to understand what happens to the cells during disease and how can they be repaired," said researcher Yan Hu, PhD.
During development, an S1P1 mutation inhibits the transition of heart muscle tissue from sponge-like to smooth and solid; instead, the tissue remains sponge-like. Additionally, bundles of muscle tissue called “trabeculations” collect in the heart, become compacted, and interfere with heart function in other ways.
The heart’s inner lining forms from the same precursor stem cells that eventually form blood cells, a finding made by past studies from Sequeira-Lopez’s lab. This, she says, highlights the plasticity of precursor cells that wasn’t understood as well by scientists in the past.
"Every organ is a complex machine built by many different cell types,” Hu said. “Knowing the origin of each cell and which genes control their normal function are the foundations for scientists to decipher the disease process and eventually to find out how to guide the cells to self-repair or even to build up a brand new organ using amended cells from the patients."
"We hope," concluded researcher Brian C. Belyea, MD, "that this is a stepping stone for our clinical colleagues."
The present study is published in the journal Scientific Reports.
Sources: Cincinnati Children’s, University of Virginia Health System