Understanding the Spread of Parkinson's from the Gut to the Brain
Multiple research studies have indicated that Parkinson’s disease, a neurodegenerative disorder impacting the brain, can begin in the gut. Scientists have now used a powerful mouse model that mimics the motor and non-motor symptoms of early- and late-stage Parkinson's disease to show how that can happen. The findings have been reported in Neuron. Learn more about Parkinson's disease from the video.
"Since this model starts in the gut, one can use it [to] study the full spectrum and time course of the pathogenesis of Parkinson's disease," explained the co-senior study author Ted Dawson, a professor of neurology at the Johns Hopkins University School of Medicine. "For instance, one could test preventive therapies at early pre-symptomatic stages of Parkinson's disease all the way to full-blown Parkinson's disease in one animal model."
In Parkinson’s, forms of an improperly folded protein called α-synuclein (described in the video) build up in the brain. Nerve cells in an area called the substantia nigra pars compacta (SNc), which make the signaling molecule dopamine, start to die. People with the disease experience motor symptoms like rigidity and tremors, and problems speaking and balancing, for example. There are no treatments that can stop the progression of the disease or relieve advanced symptoms.
Previous work has suggested that pathological α-synuclein uses the vagus nerve to reach the brain and the SNc, where the dopamine-producing neurons start to die. Only now has this been demonstrated in an animal model.
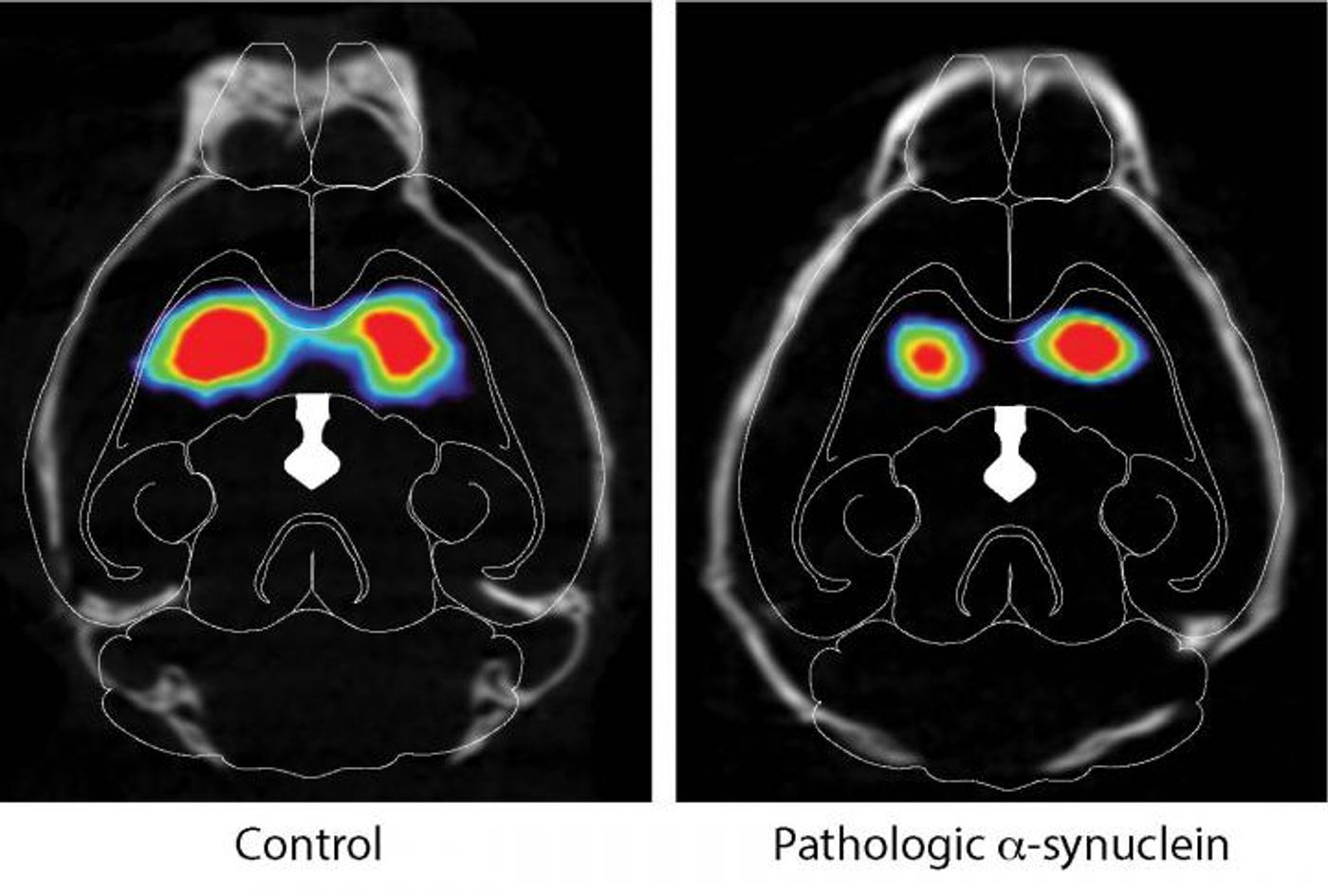
In this work, Dawson and co-senior study author Han Seok Ko of the Johns Hopkins University School of Medicine injected mouse α-synuclein preformed fibrils into gut muscles with a connection to the vagus nerve. A month later, pathologic α-synuclein had reached the brainstem’s lowest part. By three months, it had moved higher to the SNc, and had even gone beyond into other areas like the hypothalamus and prefrontal cortex. It was in even more parts of the brain seven months later, including the olfactory bulb and hypothalamus.
By then, a lot of neurons had been lost from the SNc and striatum. The injected mice also showed other symptoms including anxiety, depression, olfactory dysfunction, and cognitive deficits that disrupted spatial learning and memory and other functions.
In another group of animals that got the injection but their vagal nerve fibers were severed, no transmission of pathologic α-synuclein was observed and their were no symptoms of disease.
"There are at least three major implications of the study," said Dawson. "One: it is likely to galvanize future studies exploring the gut-brain connection. Two: it will spur investigations focused on the factors, molecules, or infections that might start the misfolding and propagation of α-synuclein. And three: it suggests that treatments could be aimed at preventing the spread of pathologic α-synuclein from the gut to brain."
It may be challenging to confirm these findings in humans, admitted the researchers. However, a certain procedure that removes part of the vagus nerve seems to reduce Parkinson's disease risk.
"Patients should not pursue vagotomies as a preventive therapy for Parkinson's disease," Dawson cautioned. "But if one could find the major initiating factor in the gut, then one could target this in a future study and show that it prevents Parkinson's disease. Patients with pathologic α-synuclein in the gastrointestinal tract would be ideal candidates for future neuroprotective studies."
The gut is closely connected to the brain; the gut has even been referred to as our second brain. There’s a massive community of microbes that resides in our guts, and scientists have been learning more about how they impact us and our brains. Researchers have also shown that microbes in the gut are involved in Parkinson’s disease. This work may help explain how the disease can start with a microbe, maybe even a pathogen that is inhaled, and reaches the gut, where the disease starts.
The scientists are continuing to pursue this work; they want to find gastrointestinal biomarkers of the disease before symptoms start, and test therapies that stop pathologic α-synuclein from spreading to the brain.
Sources: AAAS/Eurekalert! via Cell Press, Neuron