Getting to the Details of how Antipsychotic Drugs Work
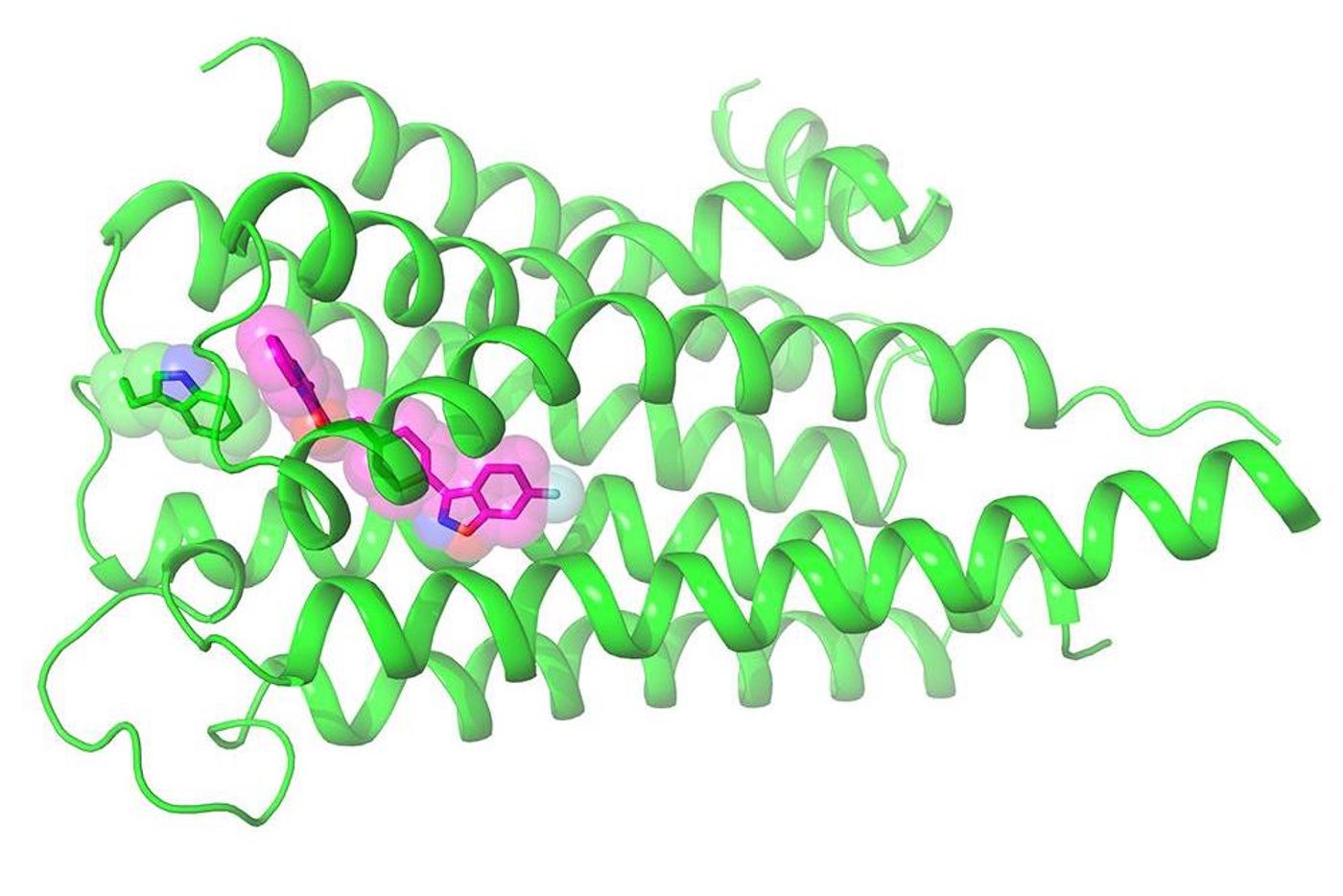
Around 1.6 percent of adults take antipsychotic medication, according to one study, but their mode of efficacy is not well understood. Individuals with conditions like bipolar disorder, schizophrenia, or who are on the autism spectrum may benefit from these drugs, but the medications can also cause serious side effects. Now, researchers at UC San Francisco and the UNC School of Medicine have revealed the crystal structure of the dopamine 2 receptor (DRD2) in high resolution as it is bound to risperidone, an antipsychotic agent. This work, reported in Nature, could go a long way toward aiding drug developers and scientists that are trying to activate DRD2 specifically. That could limit some of the many side effects, which include digestive problems, dizziness, anxiety, agitation and weight gain.
“If we want to create better medications, the first step is to see what the D2 receptor looks like in high-resolution detail when it’s bound tightly to a drug,” said senior author Bryan L. Roth, MD, PhD, the Michael Hooker Distinguished Professor of Protein Therapeutics and Translational Proteomics at the UNC School of Medicine. “We now have the structure, and we’re exploring it to find new compounds we hope can help the millions of people in need of better treatments.”
Around a third of available drugs activate molecules called G-protein coupled receptors; located on the surfaces of cells, they send signals that exert therapeutic effects. However, a lack of understanding of the intricate differences separating the various kinds of receptors means that the drugs can affect a wide swath, rather than a specific group of them. Instead of targeting only DRD2, they might also interact with serotonin, histamine, alpha-adrenergic, or other dopamine receptors. Those unintended interactions can cause severe side effects.
Risperidone is a drug commonly used by patients with schizophrenia, bipolar, and autism spectrum disorders. It is a so-called atypical antipsychotic approved for use in kids. Now scientists have determined the high-resolution structure of the drug as it is attached to DRD2.
“With this high-resolution structure in hand, we anticipate the discovery of compounds that interact with DRD2 in specific ways important for greater therapeutic actions and fewer side-effects,” Roth said.
Confirmed by the computational work of UCSF researchers Brian Shoichet, Ph.D., and Anat Levit, Ph.D., risperidone had an unpredictable mode of binding. The receptor has a previously unknown pocket; Roth and colleagues suggested it might be a good target for more selective medications.
"For the first time, we can understand precisely how atypical antipsychotic drugs bind to their primary molecular target in the human brain," explained Dr. Laurie Nadler, chief of the neuropharmacology program at the National Institute of Mental Health (NIMH), study co-funder with the National Institute of General Medical Sciences and the National Cancer Institute. "This discovery opens the way for the rational design of a new generation of antipsychotic drugs, hopefully with more desirable effects and fewer side effects."
“Before coming to UNC, I was a psychiatrist specializing in treating schizophrenia. On a daily basis, it was clear to me that medications were only modestly effective for large numbers of patients. Our lack of knowledge into how antipsychotic drugs bind to their receptors has held back progress towards creating more effective medications,” added Roth. Solving the high-resolution crystal structure of DRD2 bound to the commonly prescribed antipsychotic drug risperidone is the first step towards the creation of safer and more effective medications for schizophrenia and related disorders.”
In the video, Shoichet briefly discusses how structural data can improve biology.
Sources: AAAS/Eurekalert!, NIMH, UNC, Nature