Saving Face: Scientists Grow Jawbones in the Lab
And this ambition has progressed leaps and bounds. Just in the past year, scientists have succeeded in lab-grown retinal nerve cells, vocal cords, heart cells, and even skin that can grow hair and sweat! Now, in another scientific first, scientists at the Columbia University have succeeded in growing live bone tissues in a dish.
Though it may not seem like it, bones are complicated tissues in our bodies. They vary in size and shape, and even density. Thus when patients are stricken with diseases, like congenital deformities, or damage their bones by accidents, it can be exceedingly challenging for doctors to find and ‘install’ replacement parts.
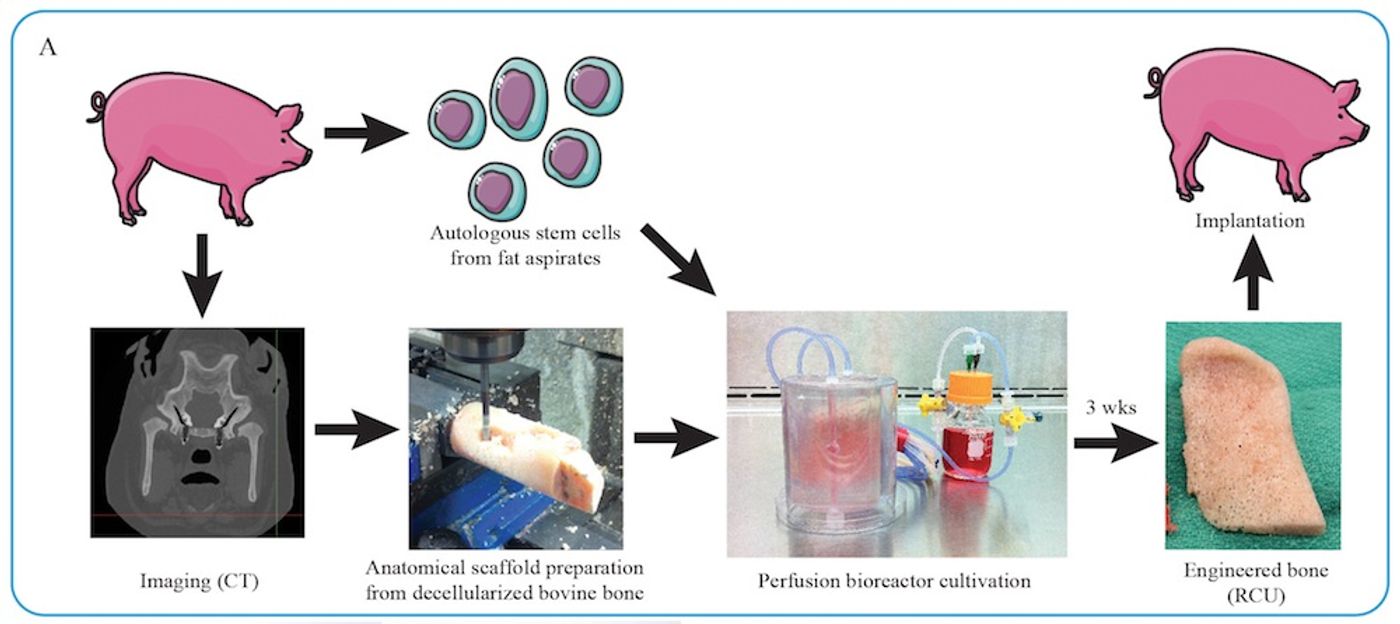
That’s where custom-engineered bones come in as the best new solution. Starting from blocks of thighbones from cows, scientists stripped the bones of cells completely. The barren scaffold was then carved into bone models that make up the lower jaw of the minipig, known as the ramus-condyle unit. They then infused these carved models with stem cells that came from the minipigs, which now lack lower jaws.
Over three weeks, the stem cells matured and made new homes in the carved bone models. In effect, the stem cells transformed the barren scaffold into living bone tissue. The grafts were then implanted back into the minipigs, chosen because their jaw anatomy and mechanics are similar to humans.
The implants helped the pigs to have use of their jaw once again. And not only that, the team observed seamless incorporation of the bone implant with the pigs’ own cells. "Unexpectedly, the lab-grown bone, when implanted, was gradually replaced by new bone formed by the body," said Gordana Vunjak-Novakovic, senior study author whose work also lead to the discovery of lab-grown heart cells. "This feature is what makes this implant your own bone that will become an integral part of the native bone."
There are additional benefits to using the recipient’s own cells to seed a scaffold into living bone tissue. First, the team observed the quality of the regenerated tissue far surpassed that of other previous attempts. Second, the team did not have to use expensive and potentially harmful drugs and growth factors to stimulate cell growth on the scaffold. And because the implant contains the recipient’s own cells, the chances of rejection are significantly reduced.
Of course, artificial solutions for bone repair and replacement also exist. However, one big disadvantage of these metal implants is the inability to produce cells in response to the recipient’s body needs. For example, titanium implants are devoid of bone marrow, which means it can’t help the recipient make new red blood cells or immune cells. Theoretically, lab-grown bone tissues with this new technique can solve this problem too.
"This is a very exciting step forward in improving regenerative medicine options for patients with craniofacial defects, and we hope to start clinical trials within a few years," said Vunjak-Novakovic.
Future clinical trials with grafts made from this technique won’t happen for a while, but the team is already looking ahead to this step. Vunjak-Novakovic has already created his own company, called epiBone, which will conduct the trials when the time comes.
Additional source: Columbia University








