Eliminating 'Toxic' RNA Molecules to Treat ALS & Dementia
Researchers have developed a way to target harmful RNA molecules that encode for disease-causing proteins. Instead of targeting the genome, this method aims for the intermediary between DNA and proteins, and could be a good way to treat diseases that are caused by the expression of mutated segments of the genome. This approach has worked to make diseased neurons healthy again in a model of dementia, and is meant to eventually be delivered to humans in a pill or an injection. The compound can also cross the blood-brain barrier. The work has been reported in the Proceedings of the National Academy of Sciences.
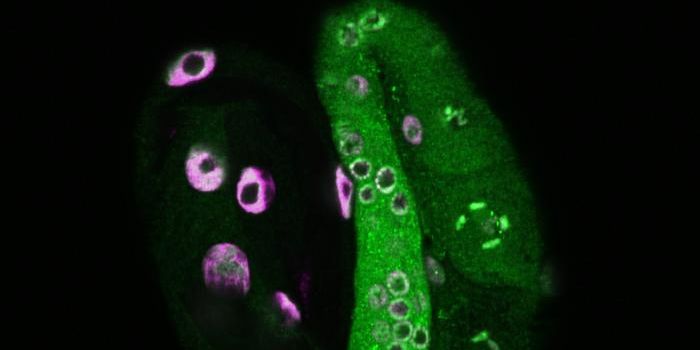
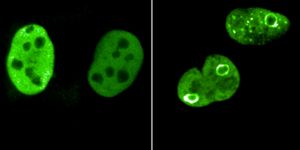
In the disorder amyotrophic lateral sclerosis (ALS), neurons that control muscles are slowly destroyed, which causes the loss and eventual death of muscles. A type of mutation in a gene called C9orf72 is a leading cause of this neurodegeneration, as well as frontotemporal dementia, a fatal disorder characterized by shrinking in the frontal and temporal lobes of the brain. This mutation is an expansion of the C9orf72 gene, in which six letters are repeated between 65 times and tens of thousands of times. These mutated sequences lead to the production of toxic proteins. In this work, the researchers created a compound that prevents the production of those toxic proteins.
"The compound works by binding to and using natural cellular processes to eliminate that disease-causing RNA by alerting the cell's degradation machinery to dispose of it as waste," explained lead inventor Matthew Disney, Ph.D., a professor and chair of the University of Florida (UF) Scripps chemistry department.
This method may also be modifiable so it can treat other diseases that involve toxic proteins.
The research team screened thousands of compounds to find 69 candidates that could disrupt the translation of the toxic RNA. They narrowed the selection down based on size, structure and other characteristics, focusing on compounds that could cross the blood-brain barrier. The final few compounds were then rated based on potency and structural simplicity, and the team selected the best drug.
Skin samples were donated by four different ALS patients, and the researchers converted cells from those samples into stem cells. The stem cells were then reprogrammed to create neurons. These patient-derived neurons were then exposed to the compound, and the researchers found that biomarkers of ALS were reduced, without causing unintended effects, revealed first study author Jessica Bush, a graduate student in the Disney lab.
"A battery of tests in neurons derived from ALS patients and in vivo models showed that compound 1 bound selectively and avidly to the toxic RNA, forcing it to be degraded by the body's own natural processes," said Bush.
In a mouse model that carried the C9orf72 mutation, daily treatments with the compound for two weeks reduced behaviors and biomarkers that are linked to ALS, and the health of the mice improved.
Now, the researchers are planning further studies to evaluate the safety and efficacy of the compound in mouse model.
"The fact that we have highlighted this in ALS shows that this can be a general approach for other neurological diseases, including Huntington's, forms of muscular dystrophy and others," added Disney.
Sources: University of Florida, Proceedings of the National Academy of Sciences