A few months ago, my fellow Labroots writer Brenda Kelley Kim did a
nice piece on
this paper from a group at Western University’s Schulich School of Medicine & Dentistry that detailed the brain changes caused by adolescent Δ-9-tetrahydrocannabinol (THC, the psychoactive ingredient in marijuana) exposure. The results were dramatic. Rats chronically exposed to THC during adolescence exhibited brain changes in adulthood that are consistent with the onset of schizophrenia and other mood disorders. Many groups have reported that adolescent THC exposure does increase risk for developing a mood disorder or psychotic-like behavior later in life, but many other groups have reported the opposite. The fact is that there is a lot of discrepancy in the literature on adolescent THC exposure. The lack of consensus could be due to differences in agonist used (THC vs synthetic cannabinoid), dosing paradigm, or age of drug administration.
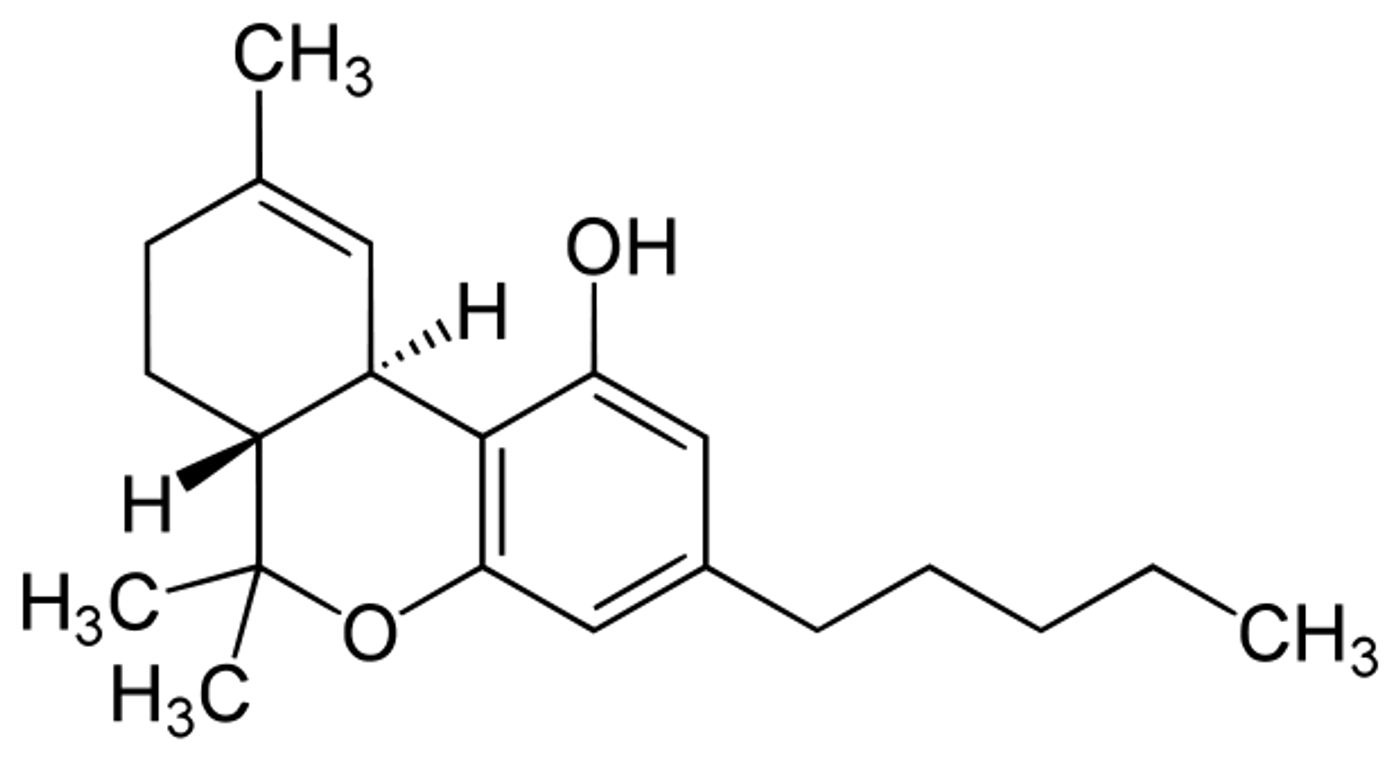
In the paper that suggests THC exposure during adolescence leads to a schizophrenic-like phenotype in adulthood, the THC dosing paradigm was aggressive and designed to elicit a behavioral response from rats. THC exposure started at postnatal day 30, right in the middle of the adolescent period in rats. Days 1-3 of THC exposure consisted of twice daily injections of 2.5 mg/kg of THC. Days 4-7 saw an increase in THC to 5 mg/kg twice a day. The last leg of THC exposure, days 8-11, were 10 mg/k of THC twice a day. Compared to this aggressive dosing paradigm, a
recent paper from Diana Dow-Edwards’s lab at SUNY Downstate Medical Center that investigated the sex- and age-specific effects of THC on rats used a much more moderate dose of THC. In their chronic THC exposure paradigm, adolescent rats were given just 3 mg/kg/day of THC for 9 days. As opposed to the researchers from Western University, the group from SUNY chose the amount of 3mg/kg/day not because it elicits drastic behavioral responses, but because the peak plasma THC levels with this dose are similar to that a human user would have after smoking 34 mg of marijuana. This is a much more physiological set-up.
The SUNY group saw that rats in the pre-pubertal period were much more sensitive to the anxiolytic and antidepressant effects of THC exposure and that, in general, male rats were more sensitive to THC. The most significant finding from this paper in relation to the paper from the Western University group is that the SUNY group did not see an effect of early adolescent THC exposure on pre-pulse inhibition in late adolescence. Pre-pulse inhibition is a behavioral measure of sensory gating, a process that is disrupted in schizophrenia and other psychotic-like disorders. The Western University group was looking at adult rats, which is a good way to look at lasting brain changes, but age of onset for schizophrenia is typically in late adolescence. What makes the SUNY group’s findings convincing is that they used a physiological amount of THC and also looked at the typical age of onset for schizophrenia and psychosis. What would have made this paper stronger would have been to test behavior at multiple time points, namely late adolescence and adulthood, and to test other aspects of a schizophrenia-like phenotype, such as dopamine activity.
There are certainly good aspects of both papers and certain areas that could have been improved, but I think it is important to look at papers on both sides of a nuanced issue, such as the long-term effects of adolescent THC exposure.