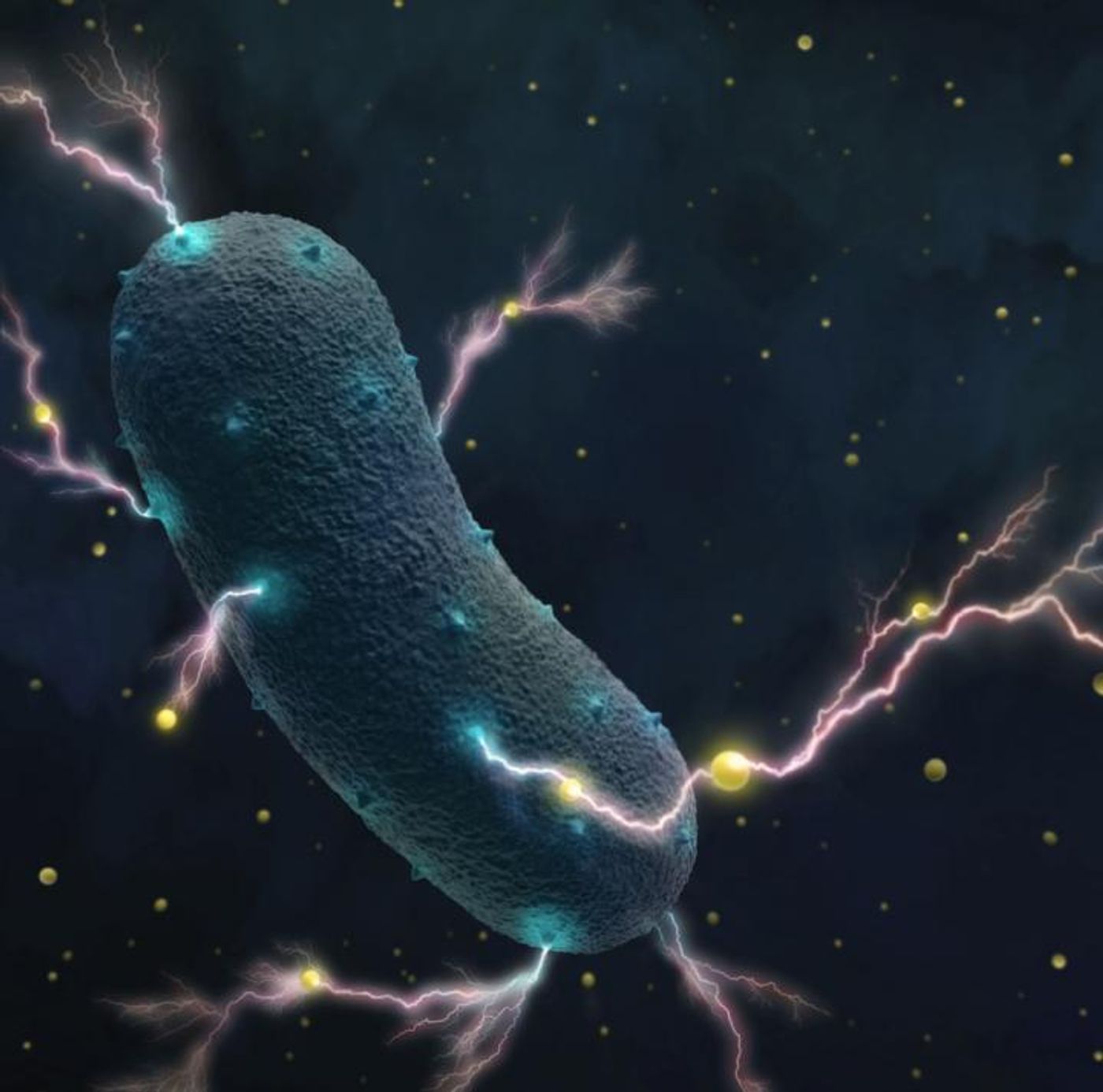
Researchers Surprised to Find Bacteria That Make Electricity in the Human Gut
We know that some bacteria can produce electricity, which was thought to be a rare characteristic of exotic microbes. But new research shows that bacteria that live in the human gut have that ability. Researchers at University of California, Berkeley determined that Listeria monocytogenes, known for causing diarrhea, has a technique for generating electricity that’s completely different from other known bacteria that also make it. These findings have been reported in Nature.
"The fact that so many bugs that interact with humans, either as pathogens or in probiotics or in our microbiota or involved in [the] fermentation of human products, are electrogenic -- that had been missed before," said Dan Portnoy, a UC Berkeley professor of molecular and cell biology and of plant and microbial biology. "It could tell us a lot about how these bacteria infect us or help us have a healthy gut."
It seems that hundreds of bacterial species produce electricity in this newly discovered way, many live in the human gut, and many can cause serious illness. The gangrene-causing bacterium Clostridium perfringens, and disease-causing Enterococcus faecalis or streptococcus make electricity as well.
Bacteria make electricity in a process that is analogous to human breathing; it removes electrons that are produced from metabolism, and aids in energy production. In plant and animal cells, mitochondria move electrons onto oxygen molecules. When bacteria live in a place where there’s no oxygen, like the human gastrointestinal tract, they have to find another molecule to accept the electrons. In some cases, it can be a metal outside the cell.
To move electrons outside the cell, a series of cascading reactions take place, creating a tiny current. Called an extracellular electron transfer chain, it could be a way to make a battery - add an electrode to a colony of these bacteria. This work could have big implications for green energy technologies; this chain is a simpler system than the one that we already knew about. It is found in microbes that only have one cell wall, called gram-positive bacteria, and which live in a flavin-rich environment (flavins are derived from vitamin B12).
"It seems that the cell structure of these bacteria and the vitamin-rich ecological niche that they occupy makes it significantly easier and more cost effective to transfer electrons out of the cell," said first author Sam Light, a postdoctoral fellow. "Thus, we think that the conventionally studied mineral-respiring bacteria are using extracellular electron transfer because it is crucial for survival, whereas these newly identified bacteria are using it because it is 'easy.'"
Light is interested in how this system is used by Lactobacillus bacteria, which are critical to cheese, yogurt and sauerkraut production. Electron transport may play a role in the taste of those foods, he suggested.
"This is a whole big part of the physiology of bacteria that people didn't realize existed, and that could be potentially manipulated," he added.
Sources: AAAS/Eurekalert! via University of California, Berkeley, Nature