A study published in Cell demonstrated how the brains of patients with different kinds of autism spectrum disorder (ASD) share common epigenetic modifications. Histone acetylation is one such type of modification, a change in a structure, a histone, which helps package DNA. 68% of patients with varying types of ASD show evidence of the same pattern of histone acetylation. This data suggests that epigenetic changes could be a common mechanism underlying a disease that has a diverse phenotype among affected individuals.
"We find epigenetic changes that are present in most patients with autism spectrum disorder, or ASD," explained a co-senior author of the work, Shyam Prabhakar of the Genome Institute of Singapore. "This suggests that, despite tremendous heterogeneity in the primary causes of autism, such as DNA mutations and environmental perturbations during development, ASD has molecular features that are commonly shared. It is encouraging that ASD has common molecular changes, because this opens up the possibility of designing drugs to correct these changes."
While it’s thought that a wide variety of genetic characteristics and environmental influences can contribute to ASD, gene mutations have not been fund to account for a large percentage of the disorder, and the investigation of structural problems with proteins have not yielded better results. Scientists have thus expanded their search for causative factors to epigenetic modifications – alterations in gene expression that are not a result of changes in the genome.
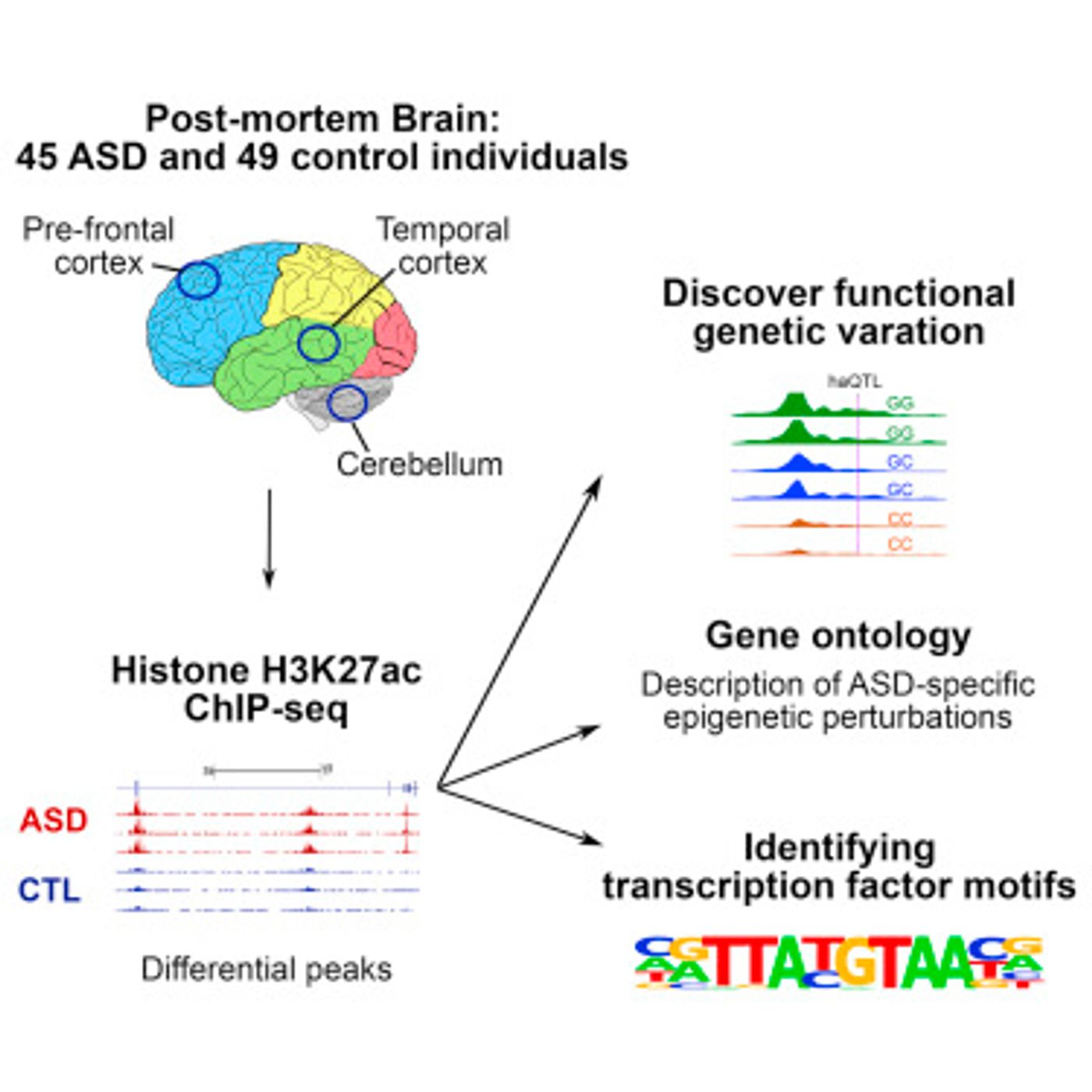
The researchers involved in this work wanted to explore modifications beyond methylation, a common epigenetic change, and looked at histone acetylation instead. The researchers, Prabhakar and co-senior study author Daniel Geschwind of UCLA, focused on an acetylation mark that has been implicated in gene activation, H3K27ac. A search for H3K27ac encompassing the entire epigenome was performed in brain tissue samples from the prefrontal cortex, temporal cortex, and cerebellar cortex, taken post-mortem from individuals with ASD and healthy control subjects that were 10 years of age and older.
They found that over 68% of ASD samples had a similar histone acetylation pattern at 5,000 gene loci, regardless of the wide range of causes and types of ASD. With the use of BrainSpan, an atlas for human brain development, they found that gene activation around 12 months after birth, when synapse formation and neuronal maturation occurs, was especially associated with increased acetylation levels in the ASD brain.
"This is the first large-scale study of how histone acetylation in the brain differs between disease and control samples, and part of a wave of new studies examining how the epigenome is perturbed in various diseases," Geschwind said. "Epigenome profiling has allowed us to see shared, unifying themes in what is often considered to be an amalgam of many different diseases rather than one single disease."
This exceptional study, made possible in large part because of advances in technology in recent years, suggests that epigenetic drugs, which are growing in use because of discoveries in cancer therapeutics, might be repurposed for use in the treatment of ASD. "Currently there are no approved drug treatments specifically for ASD, but we do hope that studies such as ours, as well as the downstream work that is surely needed, will eventually lead to new treatments," Prabhakar concluded.
The researchers would like to follow up on their results; they want to know if the epigenetic changes are causal or if they are simply associated with processes in the disease, as well as how they relate to the mechanisms of the disorder.
Check out the video above if you'd like to learn more about histone acetylation.
Sources:
AAAS/Eurekalert! via
Cell Press News,
Cell